Category
- Best Peptides for muscle growth
- Geno Pharma Domestic Warehouse 2 (Canada&USA)
- GPH(Domestic Shipping US) Warehouse 1
- Human Pharma Premium
- Phar Labs Premium-Select
- Steroids on Sale USA, Real Steroids Online
- New arrivals in USA
- Most popular steroids in USA
- Antiestrogens / Gonadotropins
- Bangkok Steroid USA
- Biopharma Steroid USA
- British Dragon
- Anabolic Steroids for Horses
- Fat-burners
- Gen Pharma USA
- Medical Pharma Steroid USA
- Medical Tech Steroid USA
- Novocrine Steroids
- HGH USA
- Omega Labs Steroid USA
- Rotterdam Steroids USA
- SARMs USA
- Sciroxx
- Sydgroup Steroid USA
- Big vetenary Steroid USA
- Watson Steroids
- XT Labs Steroids
Most Popular steroids USA
-
 Primobolan for sale 100mg 10 ml Human Pharma in USA
$115.00
Primobolan for sale 100mg 10 ml Human Pharma in USA
$115.00 -
 NPP Steroid 100mg 10 ml Premium Domestic USA
NPP Steroid 100mg 10 ml Premium Domestic USA
$99.00Original price was: $99.00.$80.00Current price is: $80.00. -
 Anavar for Sale in USA – 10mg 80 Tabs GPH-Premium
Anavar for Sale in USA – 10mg 80 Tabs GPH-Premium
$110.00Original price was: $110.00.$80.00Current price is: $80.00. -
 Buy Anavar 10mg – Purchase Geno Pharma
$99.00
Buy Anavar 10mg – Purchase Geno Pharma
$99.00 -
 Testosterone Cypionate Buy 300mg 10ml Geno Pharma
$99.00
Testosterone Cypionate Buy 300mg 10ml Geno Pharma
$99.00 -
 Buy Testosterone E 300mg 10 ml Geno Pharma Domestic USA/CA
$99.00
Buy Testosterone E 300mg 10 ml Geno Pharma Domestic USA/CA
$99.00 -
 Testosterone Cypionate 200 Biopharma 10 amp
Testosterone Cypionate 200 Biopharma 10 amp
$99.00Original price was: $99.00.$72.00Current price is: $72.00. -
 Testosterone 400 Biopharma 10 Ampoules
Testosterone 400 Biopharma 10 Ampoules
$99.00Original price was: $99.00.$75.00Current price is: $75.00. -
 Eq 300 steroid Rotterdam 10ml
Eq 300 steroid Rotterdam 10ml
$79.00Original price was: $79.00.$69.00Current price is: $69.00. -
 Steroid Deca Geno Pharma 300mg 10ml
Steroid Deca Geno Pharma 300mg 10ml
$110.00Original price was: $110.00.$99.00Current price is: $99.00. -
 Boldenone Cypionate 200 mg / 10 mL Geno Pharma
Boldenone Cypionate 200 mg / 10 mL Geno Pharma
$90.00Original price was: $90.00.$85.00Current price is: $85.00. -
 Somatrox XT Labs 150 IU x 10 vials (15 ui each)
Somatrox XT Labs 150 IU x 10 vials (15 ui each)
$450.00Original price was: $450.00.$350.00Current price is: $350.00.

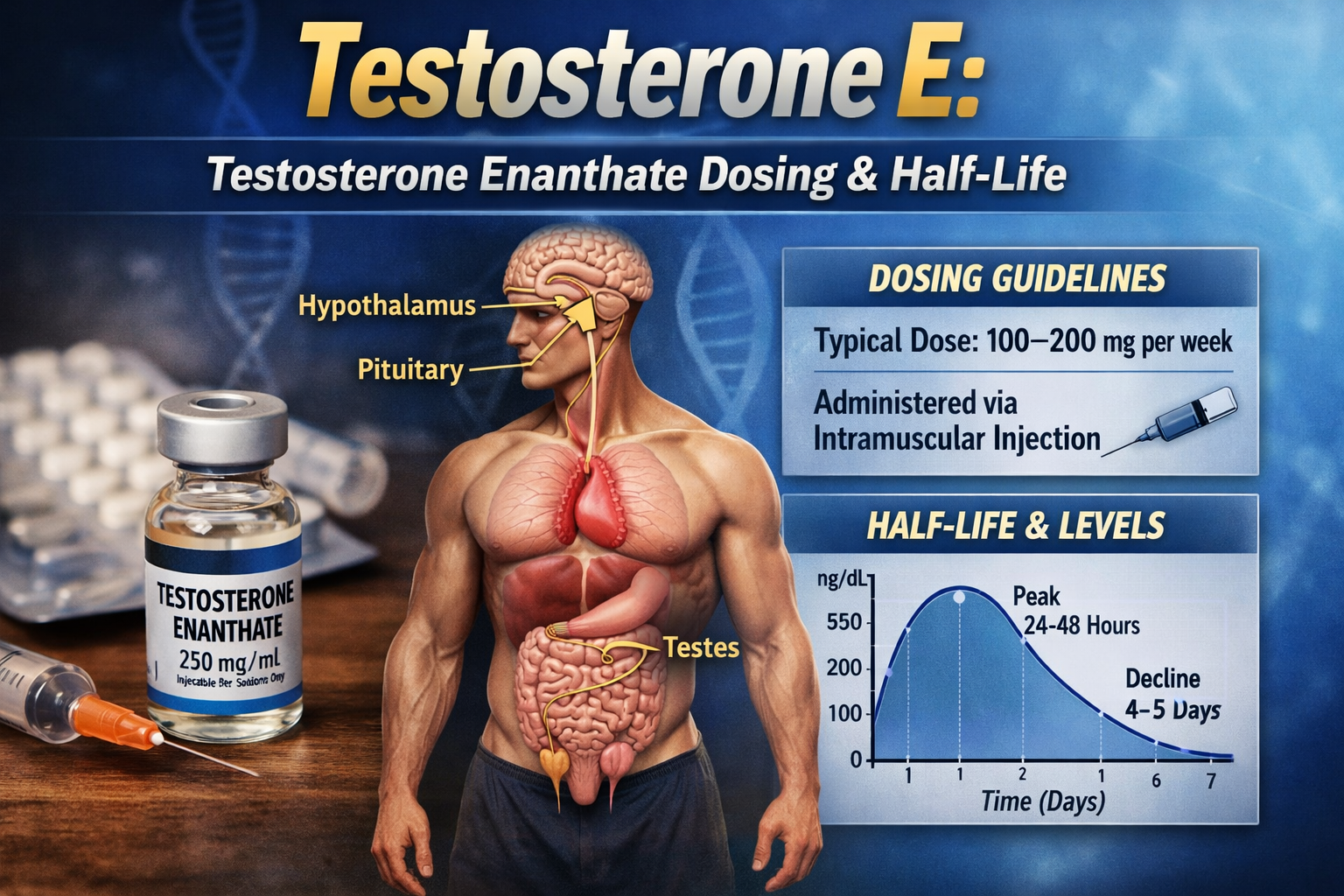
Table of Contents
ToggleWhat Is Testosterone E? Clinical Definition and Medical Context
“testosterone e” is commonly used online as shorthand for testosterone enanthate, an esterified (depot) prodrug of testosterone used clinically for testosterone replacement therapy (TRT) and other approved indications in some jurisdictions. In evidence-based practice, therapy is reserved for patients with consistent biochemical hypogonadism plus compatible symptoms/signs, and it requires structured monitoring to balance benefits and recognized risks. Major clinical guidelines emphasize confirming low testosterone with morning measurements on at least two separate occasions and evaluating etiology (e.g., primary vs secondary hypogonadism).
Testosterone esters differ mainly in pharmacokinetics (release and peak–trough behavior) rather than “different kinds of testosterone.” Testosterone enanthate and testosterone cypionate are both intermediate-acting injectable esters; differences in real-world use often reflect availability, device/formulation (e.g., subcutaneous autoinjector vs intramuscular vial), and dosing interval strategy (weekly or every-2-week regimens to reduce peaks/troughs). A Canadian regulatory monograph describes a head-to-head comparison showing similar serum testosterone profiles after equivalent “testosterone content” dosing (194 mg enanthate vs 200 mg cypionate), with peaks around days 1–2 and return to baseline by ~day 10, illustrating why shorter intervals can reduce symptomatic fluctuations.
For weekly subcutaneous testosterone enanthate autoinjector (XYOSTED), FDA labeling specifies: start 75 mg once weekly, then titrate by 25 mg based on trough total testosterone measured 7 days after the last dose after 6 weeks (target trough range commonly 350–650 ng/dL). Label pharmacokinetics show a median Tmax ~11.9 hours and mean Cmax ~790 ng/dL, Cmin ~436 ng/dL, with steady state achieved by week 6.
Safety monitoring is not optional. Well-established issues include erythrocytosis/polycythemia, blood pressure effects (notably for XYOSTED), fertility suppression, and prostate-related monitoring decisions. The EAU guideline recommends monitoring testosterone and hematocrit at 3, 6, and 12 months, then annually, with hematocrit >54% prompting dose adjustment or withdrawal (and venesection if needed).
Regarding broader benefits/risks, the large cardiovascular outcomes trial TRAVERSE (NEJM 2023)—in men 45–80 with confirmed low testosterone and elevated CV risk—found testosterone gel was noninferior to placebo for major adverse cardiovascular events (MACE), but reported higher incidence of atrial fibrillation, acute kidney injury, and pulmonary embolism in the testosterone arm. Those findings support careful patient selection and monitoring rather than indiscriminate prescribing.
Finally, many searches for “testosterone enanthate cycle” reflect non-medical anabolic-androgenic steroid (AAS) use. This article addresses the term clinically (to help clinicians counsel patients and reduce harm) but does not provide performance-enhancement dosing instructions. Testosterone (including its esters) is prohibited in sport under WADA rules without an appropriate Therapeutic Use Exemption, and non-medical use carries substantial cardiometabolic, hematologic, endocrine, fertility, and psychiatric risks.
Evidence-Based Diagnosis and Monitoring Before Prescribing Testosterone Therapy
A key principle in modern testosterone therapy is that treatment should only begin after clinical symptoms and biochemical evidence of hypogonadism are confirmed. International endocrine guidelines emphasize that testosterone replacement therapy (TRT), including formulations such as testosterone enanthate (testosterone E), must be prescribed only for men with properly diagnosed testosterone deficiency.
Because symptoms such as fatigue, low libido, reduced muscle mass, and depressed mood can have many causes, physicians must verify testosterone deficiency through repeated laboratory testing and clinical evaluation.
Confirming Low Testosterone Levels
Testosterone levels naturally fluctuate throughout the day due to circadian rhythm, with the highest concentrations typically occurring in the early morning. For this reason, medical guidelines recommend measuring total testosterone in the morning (AM sample).
However, a single abnormal result is not sufficient for diagnosis. Most endocrine guidelines require:
- Two separate morning testosterone measurements
- Laboratory confirmation of consistently low testosterone levels
- Presence of clinical symptoms consistent with hypogonadism
Only when both biochemical and clinical criteria are met should testosterone therapy be considered.
Determining the Underlying Cause
After confirming low testosterone levels, physicians investigate the underlying cause of hypogonadism. This evaluation helps distinguish between:
- Primary hypogonadism, where the testes cannot produce adequate testosterone
- Secondary hypogonadism, caused by dysfunction in the hypothalamus or pituitary gland
The diagnostic workup typically includes:
- Luteinizing hormone (LH)
- Follicle-stimulating hormone (FSH)
- Prolactin levels when clinically indicated
In some patients, clinicians may also evaluate:
- Sex hormone-binding globulin (SHBG)
- Free testosterone
These markers help determine whether total testosterone accurately reflects biologically active hormone levels.
Assessing Risks Before Starting Testosterone Therapy
Before initiating TRT with compounds such as testosterone enanthate, clinicians must evaluate potential contraindications and patient-specific risk factors.
Important considerations include:
- Fertility plans, since TRT suppresses endogenous testosterone production and sperm formation
- Baseline hematocrit, because testosterone therapy can increase red blood cell production
- Cardiovascular risk factors, including hypertension and existing heart disease
- Prostate health, typically evaluated using prostate-specific antigen (PSA) testing when appropriate
A shared decision-making process between the physician and patient ensures that the expected benefits outweigh the potential risks.
Monitoring During Testosterone Therapy
Once treatment begins, structured follow-up is essential to maintain safety and therapeutic effectiveness.
Major endocrine and urological guidelines recommend monitoring at:
- 3 months
- 6 months
- 12 months
- annually thereafter
Routine follow-up assessments may include:
- Serum testosterone levels
- Complete blood count (including hematocrit)
- Prostate-specific antigen (PSA) when indicated
- Blood pressure and cardiovascular risk markers
If hematocrit rises above 54%, or if significant adverse effects occur, clinicians may:
- adjust the dose
- extend the dosing interval
- temporarily discontinue therapy
- or perform therapeutic phlebotomy when necessary.
Careful monitoring ensures that testosterone therapy remains safe, effective, and aligned with clinical guidelines.
Clinical fundamentals
where testosterone is produced
If a reader searches “where testosterone is produced,” the clinically useful answer is: production is regulated by the hypothalamic–pituitary–gonadal axis. The pituitary secretes LH, which stimulates Leydig cells in the testes to produce testosterone; FSH mainly supports spermatogenesis in seminiferous tubules.
Quantitatively, the EAU educational appendix describes that ~5–10 mg of testosterone are secreted daily on average, with only a small extra-gonadal contribution derived from adrenal androgen precursors (e.g., DHEA) in men. It also summarizes binding: most circulating testosterone is protein-bound, with only ~1–2% free.
These physiology points matter for “testosterone e” because any exogenous testosterone (including testosterone enanthate) can suppress endogenous production through negative feedback—affecting fertility and testicular volume—and because total vs free testosterone interpretation depends on SHBG and albumin binding.
Testosterone effects
Clinically, testosterone and its active metabolite dihydrotestosterone (DHT) mediate development and maintenance of male sexual organs and secondary sex characteristics, as described in FDA labeling for testosterone products (e.g., growth/maturation of prostate and seminal vesicles, hair distribution, and body composition changes).
At the systems level, guidelines and trials link appropriately prescribed TRT to improvements in domains that are clinically meaningful for selected patients (sexual function, anemia, bone density), while still emphasizing uncertainty about long-term outcomes in some populations and the need for individualized decisions.
What testosterone does to men
Testosterone plays a central role in male physiology by supporting sexual development, libido, muscle protein synthesis, bone density, red blood cell production, mood, and energy levels. In adolescence, it helps drive changes such as increased muscle mass, facial and body hair growth, and deepening of the voice, while in adulthood it continues to influence body composition, sexual function, and overall metabolic health. Because of these wide-ranging effects, low testosterone can contribute to symptoms such as fatigue, reduced libido, depressed mood, and loss of lean mass.
Diagnosis and treatment eligibility
Evidence-based practice requires that “low testosterone” is not diagnosed from symptoms alone. The AUA guideline uses total testosterone <300 ng/dL as a reasonable biochemical cutoff and states diagnosis should be made after two early-morning total testosterone measurements on separate occasions.
EAU guidance similarly emphasizes morning, fasting measurement (typically 07:00–11:00) with confirmatory testing before treatment; it notes that positive outcomes are more consistently documented at lower testosterone levels and proposes thresholds such as 12 nmol/L in symptomatic men (with higher certainty in more severe deficiency).
FDA labels for injectable testosterone products likewise direct clinicians to confirm hypogonadism with morning testosterone on two separate days before initiation.
Testosterone E in practice
What “testosterone e” means clinically
In clinical documentation, patients may say “testosterone e,” “testosterone enanthate,” or “test enanthate.” Clinically, these refer to testosterone delivered as the enanthate ester, designed to slow release from the injection depot (intramuscular oil-based preparations or specific subcutaneous devices).
A practical implication is that “half-life” and “duration” depend on formulation (IM oil depot vs SC autoinjector), patient factors, and dosing interval; peak-to-trough volatility is often a driver of symptoms and adverse effects (e.g., erythrocytosis risk is higher with parenteral vs topical formulations).
Testosterone enanthate half life
The keyword “testosterone enanthate half life” is tricky because different sources may report:
- the biological half-life of free testosterone (minutes to hours), and
- the effective half-life/duration of a depot ester formulation (days), which is often “absorption-limited.”
A clinically useful way to frame this is: the depot injection can maintain physiologic or supraphysiologic testosterone levels for many days, often requiring weekly or biweekly dosing to reduce fluctuation. A Canadian monograph describing pharmacokinetics of testosterone enanthate vs cypionate reports serum testosterone rises sharply and peaks around days 1–2, then declines toward baseline by about day 10 after a specific equivalence-dosed comparison—consistent with the clinical observation that symptoms can fluctuate when dosing intervals are long.
For subcutaneous testosterone enanthate autoinjector (XYOSTED), the FDA label provides pharmacokinetic parameters across the 7‑day interval at steady state: mean Cmax ~790 ng/dL, Cmin ~436 ng/dL, and median Tmax ~11.9 hours, showing a controlled peak and a trough remaining within the physiologic range for most patients under labeled use.
Suggested pharmacokinetic chart (for the article page): An overlay of concentration–time curves for (a) weekly SC testosterone enanthate autoinjector vs (b) IM testosterone enanthate/cypionate every 2–4 weeks, highlighting peak-to-trough ratios. The Andrology review notes that weekly SC testosterone enanthate has a Cmax/Cmin ratio ~1.8, whereas IM testosterone enanthate/cypionate regimens may reach peak-to-trough ratios ~2 to 5.3 depending on dose and schedule.
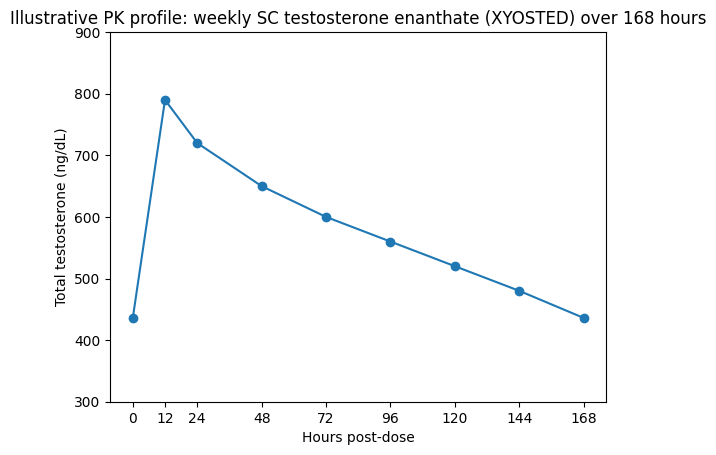
Illustrative PK profile: weekly SC testosterone enanthate (XYOSTED) over 168 hours01224487296120144168Hours post-dose900850800750700650600550500450400350300Total testosterone (ng/dL)Mostrar código
Chart values are illustrative to visualize peak-to-trough behavior and are anchored to FDA label parameters (Cmin and Cmax with Tmax) rather than patient-level raw timepoints; real profiles vary across individuals.
Testosterone enanthate dosage
When clinicians discuss testosterone enanthate dosage, the key is aligning dose and interval to (a) symptom response and (b) measured testosterone levels (ideally minimizing supraphysiologic peaks and subtherapeutic troughs), while (c) monitoring hematocrit and other safety parameters.
Guideline-based regimens often favor weekly or every-2-week schedules for short-/intermediate-acting injectables:
- The Society for Endocrinology guideline table lists testosterone enanthate or cypionate 150–200 mg IM every 2 weeks or 50–100 mg IM or SC once weekly (as examples of short-acting injections), explicitly acknowledging peak–trough symptoms and polycythemia risk with injectables.
- In contrast, some FDA labeling for testosterone cypionate allows a broader range (50–400 mg every 2–4 weeks) and emphasizes individualization—yet longer intervals can amplify peaks and troughs in practice, so many clinicians fractionate dosing when consistent with local guidance and patient needs.
Dose and schedule table for TRT and clinically supervised use
| Clinical context | Formulation | Typical regimen examples (adults) | How to time testosterone blood draw for titration | Evidence/guideline basis |
|---|---|---|---|---|
| TRT (adult male hypogonadism) | IM/SC testosterone enanthate or testosterone cypionate | 50–100 mg weekly IM/SC or 150–200 mg IM every 2 weeks (examples used in clinical guidance to reduce peak–trough symptoms) | Use trough levels (just before next injection) to adjust interval/dose; some protocols use mid‑interval sampling depending on regimen | Society for Endocrinology guidance table and discussion of peak–trough issues |
| TRT (adult male hypogonadism) | Weekly SC testosterone enanthate autoinjector (XYOSTED) | Start 75 mg SC weekly; adjust by 25 mg to 50 mg or 100 mg based on trough | Measure total testosterone trough 7 days after last dose after 6 weeks (and 6 weeks after changes); trough ~350–650 ng/dL generally targeted | FDA label dosing and titration rules |
| TRT (label range, not necessarily optimal for symptom stability) | IM testosterone cypionate (labeling) | 50–400 mg every 2–4 weeks (deep IM gluteal) | Labeling does not specify “fractionated” strategy; clinicians often monitor and individualize to avoid peaks/troughs | FDA label for testosterone cypionate |
| Non-medical “performance enhancement” | Any testosterone ester (“cycle”) | No medically endorsed dose or ‘cycle’ exists for performance enhancement; risks rise with supraphysiologic dosing and polypharmacy | Clinically, treat as a counseling and risk-management problem; evaluate for complications and substance use disorder | WADA prohibits anabolic agents/esters; FDA label warns about abuse; guidelines focus on indicated TRT only |
Testosterone enanthate benefits
The keyword “testosterone enanthate benefits” should be handled carefully: clinically, benefits are expected from restoring testosterone to physiologic ranges in appropriately diagnosed patients, and those benefits are not unique to enanthate versus other formulations—differences are mostly practical and pharmacokinetic.
A high-quality individual participant data meta-analysis (Lancet Healthy Longevity, 2023) in men with baseline total testosterone <12 nmol/L found testosterone treatment improved erectile function scores versus placebo, reaching a minimal clinically important difference for mild erectile dysfunction; it also improved Aging Males’ Symptoms score and some quality-of-life subscores, though it did not significantly improve depression scores in that analysis.
TRAVERSE (NEJM 2023) also summarizes that controlled trials have shown improvements in sexual function, bone mineral density, and anemia correction in older men—providing context for expected physiologic benefits when used appropriately.
For older men in particular, the EMAS 2023 position statement recommends TRT only for symptomatic men with confirmed low testosterone after discussing uncertainties, and notes TRT may be considered for specific problems (e.g., sexual dysfunction, severe insulin resistance/prediabetes, osteoporosis in combination with proven strategies, selected depressive symptoms), while discouraging routine use for physical function or cognition.
testosterone enanthate cycle
The search phrase “testosterone enanthate cycle” is widely used outside medicine to describe time-limited supraphysiologic AAS use for physique/performance. In evidence-based clinical endocrinology/urology, testosterone is prescribed as replacement for confirmed deficiency—not as “cycling”—and dosing is adjusted to keep testosterone levels within the physiologic range while minimizing harms.
Because manufacturers and regulators explicitly warn that testosterone can be abused at higher than prescribed doses and in combination with other anabolic agents, clinicians should treat “cycle” language as a signal to assess: cardiovascular risk, hematocrit/erythrocytosis risk, blood pressure, psychiatric symptoms, fertility intentions, and potential withdrawal/hypogonadism on cessation.
Enanthate vs cypionate in clinical decision-making
This section is designed to satisfy searches for testosterone enanthate vs cypionate and testosterone enanthate vs cypionate dosage.
Core takeaway
Testosterone enanthate and testosterone cypionate deliver the same active hormone (testosterone) after ester cleavage; differences are small compared with the effect of dose, injection interval, and delivery system on peaks/troughs and adverse effects.
A regulatory monograph notes that dosing can be made “equivalent” by matching unesterified testosterone content (e.g., 194 mg enanthate vs 200 mg cypionate), and under those conditions serum testosterone levels were described as identical, peaking in days 1–2 and returning toward baseline by day 10—supporting the practical view that these esters are clinically similar when dosed appropriately.
In clinical practice, “enanthate vs cypionate” decisions often revolve around: regional availability, insurance coverage, device preference (self-injection), and tolerability—especially managing peak-to-trough variability and erythrocytosis risk. The EAU guideline notes hematocrit elevations are more common with parenteral formulations, underscoring that formulation/route can matter as much as ester choice.
Comparison table: testosterone enanthate vs testosterone cypionate
| Domain | Testosterone enanthate | Testosterone cypionate | Practical clinical note | Key sources |
|---|---|---|---|---|
| Active hormone | Testosterone (after ester cleavage) | Testosterone (after ester cleavage) | Clinical effect is from testosterone; ester mainly changes release profile | XYOSTED label notes enanthate → testosterone via ester cleavage |
| Common routes | IM oil depot; also SC in some regimens/devices | IM oil depot; SC used in some practices | Route, needle burden, and patient self-administration drive adherence | Society for Endocrinology discusses IM/SC options for enanthate or cypionate |
| Pharmacokinetics (concept) | Intermediate-acting injectable; peaks in first days, declines over ~1–2 weeks depending on regimen | Intermediate-acting injectable; similar pattern | Dose interval and fractionation reduce peaks/troughs and may reduce erythrocytosis risk | Head-to-head equivalence described in Health Canada monograph |
| Half-life / duration signal | Often described as multi-day depot exposure; clinically relevant “coverage” can be ~10+ days in some regimens | Label cites IM half-life ~8 days for cypionate | Depot kinetics are absorption-limited; “half-life” depends on model; focus on measured troughs | Cypionate half-life cited in labeling material |
| Dosing (TRT examples) | 50–100 mg weekly or 150–200 mg every 2 weeks (example regimens) | Same example regimens | “testosterone enanthate vs cypionate dosage” is often essentially the same in TRT protocols; adjust to labs/symptoms | Society for Endocrinology dosing examples |
| Availability (US example) | XYOSTED weekly SC autoinjector exists; IM vials exist | Multiple IM cypionate products exist | Device availability can change adherence and peak–trough profile | XYOSTED dosing/PK and warnings |
| Side effects (overlap) | Shared androgenic AEs; erythrocytosis, fertility suppression, acne, mood changes possible | Shared androgenic AEs; cypionate labeling includes infertility warning; half-life statement | Specific AE rates depend on dose/interval and patient factors; injectable peaks can worsen Hct/E2 | Cypionate and XYOSTED labeling + EAU monitoring threshold |
Safety, monitoring, and adverse effects
testosterone enanthate side effects
The phrase “testosterone enanthate side effects” should be presented in a risk-stratified way:
Common, clinically relevant adverse effects across testosterone formulations include acne/oily skin, fluid retention, gynecomastia (via aromatization), mood/behavior changes, and injection-site reactions. Labels also highlight more serious risks: polycythemia/erythrocytosis, venous thromboembolism signals, blood pressure increases (particularly emphasized for XYOSTED), potential worsening of BPH symptoms, and suppression of spermatogenesis at higher doses.
XYOSTED-specific label signals:
In a 6‑month study, adverse reactions ≥2% included hematocrit increased (8.3%) and several injection-site events (e.g., hemorrhage, bruising).
The label also states clinicians should monitor blood pressure and that uncontrolled hypertension is a reason to avoid use; it warns about abuse and notes high doses can lower sperm count.
Safety monitoring checklist and recommended lab tests
A practical monitoring plan should align with major guidelines while adapting to formulation and patient risk.
Baseline (before starting TRT):
Confirm diagnosis with two morning testosterone levels; evaluate etiology and comorbidities; discuss fertility.
At minimum, document: hematocrit/hemoglobin, blood pressure and CV risk profile, and a prostate cancer risk discussion/assessment appropriate to age and risk factors (shared decision-making).
Follow-up cadence (common guideline convergence):
EAU recommends checking testosterone and hematocrit at 3, 6, and 12 months and annually thereafter, and specifies that hematocrit >54% requires adjustment or withdrawal (and venesection if required).
Endocrine Society guidance includes monitoring testosterone and hematocrit at 3–6 months, again at 12 months, then annually, with prostate risk reassessment during the first year for those choosing monitoring.
EMAS 2023 similarly recommends monitoring at 3, 6, 12 months, then yearly, including total testosterone, hematocrit, and PSA, and notes PSA rise thresholds prompting urologic evaluation (e.g., >1.4 ng/dL rise within 3–12 months).
Monitoring table (clinic-ready)
| Timepoint | What to assess | Labs/tests (typical) | Action thresholds (examples from guidelines/labels) |
|---|---|---|---|
| Baseline | Symptoms, infertility intentions, comorbidities; CV risk; prostate risk discussion | Total testosterone (AM) ×2; CBC (Hgb/Hct); ±LH/FSH/prolactin; PSA per age/risk/shared decision; BP | Delay/avoid TRT in inappropriate indications; document counseling |
| 3 months | Response + adverse effects; injection technique if relevant | Total testosterone (timed to regimen); CBC; BP (especially XYOSTED); ±PSA per plan | Rising Hct requires dose/interval adjustment; high-risk patients may need closer follow-up |
| 6 months | Continue titration to stable range; reassess adherence | Testosterone + CBC; BP; symptom scales | For XYOSTED, trough-guided titration after 6 weeks and periodically |
| 12 months | Confirm durable benefit; reassess ongoing indication | Testosterone + CBC; PSA per shared decision; cardiometabolic review | Hct >54% → adjust/withdraw; consider venesection if needed |
| Yearly (or more often if high risk) | Long-term benefit vs risk | Testosterone + CBC; PSA/BPH symptom review as planned; BP | Continue only if benefit outweighs risk for the individual patient |
Special risk domains clinicians should address
Erythrocytosis / polycythemia:
EAU cites elevated hematocrit risk particularly with parenteral formulations and recommends maintaining hematocrit below critical thresholds; it explicitly states hematocrit >54% requires therapy adjustment or withdrawal (and venesection if required).
Blood pressure and cardiovascular risk:
XYOSTED labeling highlights blood pressure increases and recommends periodic monitoring; uncontrolled hypertension is a key caution.
TRAVERSE found no increase in MACE compared with placebo (noninferiority met), but did observe higher rates of atrial fibrillation, acute kidney injury, and pulmonary embolism—important for counseling and surveillance in higher-risk men.
Prostate monitoring:
A JCEM 2024 expert review synthesizing TRAVERSE and other randomized data concludes adverse prostate events were low in carefully screened men and supports baseline prostate risk evaluation with a standardized monitoring plan to minimize unnecessary biopsies while detecting high-grade cancers.
A linked randomized trial report (JAMA Network Open) similarly describes low rates of adverse prostate events in appropriately selected men with PSA thresholds and verification procedures to mitigate ascertainment bias.
Fertility and spermatogenesis:
Exogenous androgens suppress endogenous testosterone production (negative feedback) and at high doses may suppress spermatogenesis; labeling explicitly notes sperm count reduction at high doses.
EMAS explicitly states TRT is contraindicated in hypogonadal men actively seeking fertility treatment.
Visual: diagnosis-to-monitoring workflow
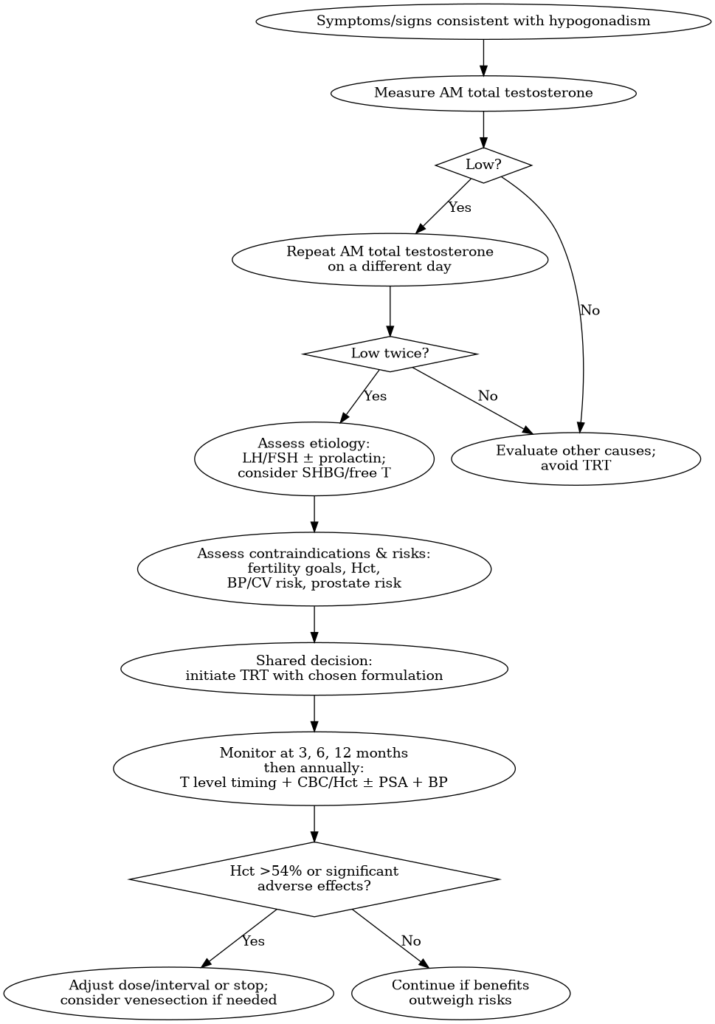
Workflow reflects common elements across EAU/Endocrine Society/EMAS and FDA labeling.
Risk–benefit synthesis, clinical recommendations, and prioritized sources
Risk–benefit analysis in 2020–2026 evidence context
Benefits are most defensible when: (1) biochemical hypogonadism is clearly documented, (2) symptoms are plausibly attributable to low testosterone, and (3) monitoring is implemented. The 2023 IPD meta-analysis supports meaningful improvement in mild erectile dysfunction and some symptom scales in men with low baseline testosterone (<12 nmol/L), with heterogeneity in other domains such as depression.
In older, higher-cardiovascular-risk men, TRAVERSE provides the strongest randomized evidence base to date that TRT (gel, titrated to 350–750 ng/dL) is not associated with higher MACE than placebo over a mean ~22 months, while still flagging safety signals (AF, AKI, PE) that justify careful surveillance.
For prostate outcomes, contemporary randomized evidence suggests that in men screened to exclude high baseline prostate cancer risk, serious prostate events are uncommon, but PSA rises can increase biopsy cascade risk; thus standardized monitoring and shared decision-making are central.
The most consistent and actionable harm signal across guidelines is hematocrit elevation: EAU recommends hematocrit monitoring at 3/6/12 months and annually, with >54% requiring dose adjustment or withdrawal. This single parameter is a major lever clinicians can use to reduce thrombotic risk.
Clinical recommendations for “testosterone e” prescribing (evidence-based, neutral)
- Do not prescribe testosterone e (testosterone enanthate) for non-specific symptoms without confirmed biochemical deficiency. AUA and EAU emphasize diagnosis based on symptoms plus consistently low levels, confirmed with repeat AM testing.
- Choose regimen to minimize peaks/troughs and monitor objectively. Short-acting injections can create “peak & trough” symptoms and polycythemia risk; weekly or biweekly dosing strategies are commonly used in clinical guidance to reduce volatility.
- Use label-specific titration rules when using device-specific testosterone enanthate products (e.g., XYOSTED). Follow trough-guided titration after 6 weeks and periodically, per FDA labeling.
- Treat hematocrit as a high-priority safety endpoint. Monitor as scheduled; intervene at >54% with adjustment/withdrawal and consider venesection if required.
- Integrate CV and renal risk discussion, especially in high-risk patients. TRAVERSE supports MACE noninferiority but identifies specific adverse event increases; counsel and monitor accordingly.
- Be explicit about fertility suppression. Avoid TRT in men actively seeking fertility treatment (EMAS) and counsel all patients that exogenous testosterone can suppress spermatogenesis.
- Address “cycle” language as a clinical red flag, not a treatment plan. Testosterone is prohibited in sport without TUE (WADA), and labeling warns about abuse; clinicians should focus on harm reduction, screening for complications, and offering evidence-based support rather than providing dosing instructions for non-medical use.
Prioritized sources for citation and further reading
Regulatory and official documents (highest authority for product-specific dosing/warnings):
- FDA Prescribing Information: XYOSTED (testosterone enanthate) 2025 label (dose titration, BP warning, PK parameters, abuse warning).
- FDA Prescribing Information: Testosterone cypionate injection 2022 label (diagnostic confirmation, labeled dosing range).
- Health Canada Product Monograph: Testosterone Enanthate Injection (enanthate vs cypionate equivalence study; PK narrative).
- WADA International Standard Prohibited List 2026 (testosterone and its esters prohibited in sport).
Clinical guidelines and position statements (practice standards):
- EAU Guidelines on Sexual & Reproductive Health (male hypogonadism chapter): diagnostic timing, thresholds, hematocrit monitoring and >54% action.
- EAU Appendix: Physiology of testosterone production (clear explanation of LH/FSH axis, production amounts, binding).
- Endocrine Society Clinical Practice Guideline (JCEM 2018): monitoring schedule and prostate monitoring triggers (still widely referenced).
- Society for Endocrinology guideline (Clinical Endocrinology 2022; accepted version): practical formulation table including enanthate/cypionate dosing examples and monitoring concepts.
- EMAS 2023 position statement (Maturitas): indications/contraindications in older men and monitoring schedule; cautions post-MI/stroke and fertility.
Key peer-reviewed studies and syntheses (2020–2026 prioritized):
- TRAVERSE trial (NEJM 2023): cardiovascular safety outcomes, adverse event signals.
- Lancet Healthy Longevity 2023 IPD meta-analysis: symptomatic benefits (IIEF-15 improvements; AMS score).
- JCEM 2024 review on prostate risk/monitoring during TRT (TRAVERSE-informed): practical baseline evaluation and monitoring framework.
- JAMA Network Open trial report on prostate safety events during TRT (screened population; PSA thresholds).
- Andrology review (PK profiles vs diurnal variation; includes SC testosterone enanthate and peak-to-trough comparisons): useful for explaining why weekly SC TE has lower peak-to-trough ratio than many IM schedules.
Closing clinical note
“testosterone e” content performs best (SEO + clinical integrity) when it answers what patients ask—dose, half-life, effects, enanthate vs cypionate—while consistently reinforcing the medical standard: confirm diagnosis, treat to physiologic targets, and monitor hematocrit, blood pressure, and prostate risk using guideline-based schedules.
Testosterone E FAQ: Dosage, Benefits, Side Effects and Therapy Questions
Best testosterone e supplements available for men
Testosterone enanthate (often referred to online as “testosterone e”) is a prescription medication used in testosterone replacement therapy (TRT). It is not considered a dietary supplement. Products marketed as “testosterone boosters” are typically over-the-counter supplements that aim to support hormone health through ingredients such as zinc, vitamin D, or herbal extracts. However, these products are not equivalent to prescription testosterone therapy and generally do not raise testosterone levels to the same extent as medically prescribed treatments.
Men experiencing symptoms of low testosterone should consult a qualified healthcare provider rather than relying solely on supplements marketed online.
What is testosterone enanthate used for?
Testosterone enanthate is a long-acting injectable form of testosterone used primarily for testosterone replacement therapy (TRT) in men diagnosed with hypogonadism. Hypogonadism occurs when the body produces insufficient testosterone due to dysfunction of the testes or the hypothalamic-pituitary system.
Medical testosterone therapy may help restore normal hormone levels and improve symptoms such as low libido, fatigue, decreased muscle mass, and reduced bone density.
Testosterone e therapy clinics near me
Many regions have specialized men’s health clinics, endocrinology practices, and urology clinics that evaluate and treat low testosterone. These clinics typically offer diagnostic testing, hormone evaluations, and medically supervised testosterone replacement therapy.
Patients searching for testosterone therapy clinics should ensure that providers are licensed medical professionals who perform proper hormone testing before prescribing treatment.
How does testosterone replacement therapy work?
Testosterone replacement therapy works by restoring testosterone levels in individuals who have clinically confirmed testosterone deficiency. Injectable forms such as testosterone enanthate release testosterone slowly after injection, allowing the hormone to enter the bloodstream over several days.
Once circulating in the body, testosterone binds to androgen receptors in tissues such as muscle, bone, and the brain, regulating processes like muscle protein synthesis, red blood cell production, metabolism, and sexual function.
How to buy testosterone e online safely
Testosterone enanthate is a prescription medication in many countries. The safest way to obtain it is through a licensed healthcare provider who performs proper medical evaluation and laboratory testing.
Patients should avoid purchasing injectable hormones from unregulated online sources because these products may be counterfeit, contaminated, or improperly dosed.
What are the benefits of prescribed testosterone injections?
When prescribed for medically diagnosed testosterone deficiency, testosterone injections may help improve:
- Libido and sexual function
- Energy levels and fatigue
- Lean muscle mass and strength
- Bone mineral density
- Mood and overall well-being
Benefits vary among individuals and are most evident in patients with confirmed hypogonadism.
Top-rated testosterone e replacement products
Several testosterone replacement therapy formulations exist, including injectable testosterone esters such as testosterone enanthate and testosterone cypionate. Other options include topical gels, patches, and long-acting injectable preparations.
The best option depends on patient preference, medical history, and physician recommendations.
Common side effects of HRT with testosterone.
Common side effects associated with testosterone therapy may include increased red blood cell count, acne, fluid retention, and potential changes in cholesterol levels. Some patients may also experience suppression of natural testosterone production.
Because of these risks, physicians typically monitor hormone levels, blood counts, and other biomarkers during therapy.
Testosterone e dosage recommendations for beginners
Dosage for testosterone enanthate in testosterone replacement therapy typically ranges between 50 mg and 200 mg per week, depending on the patient’s baseline hormone levels and clinical needs.
Dosage should always be determined by a healthcare professional and adjusted based on laboratory results and treatment response.
How often are testosterone enanthate injections typically administered?
Testosterone enanthate injections are commonly administered once every one to two weeks in clinical practice. Some treatment protocols involve smaller weekly injections to maintain more stable testosterone levels.
Injection frequency depends on physician guidance and individual patient response.
Reviews of testosterone e injections for muscle growth
Some individuals report improvements in muscle mass and strength while using testosterone therapy, particularly when combined with resistance training and proper nutrition. However, testosterone therapy is medically indicated for patients with low testosterone levels and should not be used without medical supervision.
Clinical outcomes vary depending on hormone levels, lifestyle factors, and treatment protocols.
Where can I find a doctor specializing in low testosterone treatment?
Doctors specializing in testosterone deficiency treatment typically include endocrinologists, urologists, and physicians working in men’s health clinics. These specialists can evaluate symptoms, perform hormone testing, and determine whether testosterone therapy is appropriate.
Patients can often locate qualified providers through hospital networks, medical directories, or referrals from primary care physicians.
Testosterone e side effects and how to minimize them
Potential side effects of testosterone therapy may include elevated hematocrit, acne, fluid retention, and changes in cholesterol levels. These risks can often be minimized through proper dosing, regular laboratory monitoring, and follow-up appointments with a healthcare provider.
Adjusting injection frequency or dosage may also help reduce adverse effects.
What are the steps to get a prescription for testosterone therapy?
Obtaining a prescription for testosterone therapy typically involves:
- Medical consultation with a licensed healthcare provider
- Blood tests measuring testosterone levels
- Confirmation with repeat testing if levels are low
- Evaluation of potential causes of testosterone deficiency
If hypogonadism is confirmed, a physician may recommend an appropriate testosterone therapy protocol.
Where to find testosterone e gel in the US
Topical testosterone gels are available in the United States through prescription medications approved by regulatory agencies. These products are typically dispensed through licensed pharmacies after a healthcare provider evaluates the patient and prescribes treatment.
Patients should consult a physician to determine whether injectable or topical testosterone formulations are appropriate.
Online clinics offering testosterone replacement services.
Some telemedicine clinics and men’s health providers offer remote consultations for testosterone therapy. These services may include online medical evaluations, laboratory testing, and physician-guided treatment plans.
Patients should ensure that online clinics operate legally and employ licensed medical professionals.
Cost comparison of testosterone e therapy providers
The cost of testosterone therapy varies depending on the provider, treatment formulation, laboratory testing requirements, and follow-up care. Some clinics offer bundled programs that include consultations, lab testing, and medication.
Insurance coverage may reduce overall costs in cases of medically diagnosed hypogonadism.
Cost of testosterone enanthate without insurance.
Without insurance coverage, the cost of testosterone enanthate may vary depending on the pharmacy, dosage, and geographic location. Generic formulations are often less expensive than brand-name versions.
Patients may also incur additional costs for laboratory testing and physician consultations.
Testosterone e vs other testosterone replacement options
Testosterone enanthate is one of several forms of testosterone replacement therapy. Other options include testosterone cypionate injections, topical gels, transdermal patches, and long-acting injectable formulations.
Each option differs in dosing frequency, absorption characteristics, and convenience.
Is testosterone therapy covered by health insurance plans?
In many healthcare systems, testosterone therapy is covered by insurance when prescribed for medically diagnosed hypogonadism. Coverage policies vary by provider and plan.
Patients should check with their insurance company to confirm eligibility and coverage requirements.
How to start testosterone e therapy with a licensed provider
Starting testosterone therapy typically begins with a medical consultation, hormone testing, and evaluation of symptoms. If testosterone deficiency is confirmed, the healthcare provider will recommend a treatment plan and monitor the patient during therapy.
Regular follow-up visits and laboratory testing are important for maintaining safety and treatment effectiveness.
What are the different brands of injectable testosterone available?
Several pharmaceutical manufacturers produce injectable testosterone formulations. These include testosterone enanthate and testosterone cypionate products distributed under various brand and generic names depending on the country.
Availability varies by regulatory approval and regional pharmaceutical markets.
Testosterone e prescription requirements in the US
In the United States, testosterone enanthate is classified as a prescription medication and a controlled substance. This means it can only be legally obtained with a valid prescription from a licensed healthcare provider.
Patients must undergo medical evaluation and diagnostic testing before receiving treatment.
Where to buy needles and syringes for testosterone injections?
Needles and syringes used for medical injections are typically available through pharmacies, medical supply stores, or healthcare providers. Regulations vary depending on local laws, and some areas may require a prescription.
Patients should always follow safe injection practices and guidance from healthcare professionals.
What are the benefits of testosterone E?
Testosterone E (testosterone enanthate) is used in clinical practice to restore testosterone levels in men with confirmed hypogonadism. When prescribed appropriately, it may help improve libido, energy levels, lean muscle mass, bone density, and overall mood. These benefits are primarily observed in individuals with medically diagnosed low testosterone rather than in those with normal baseline levels.
How long does it take for testosterone E to kick in?
Testosterone E does not produce immediate effects. Blood testosterone levels typically rise within hours after injection, with peak levels occurring within the first 24–48 hours. However, noticeable clinical effects such as improved energy, libido, or mood may take several weeks, while changes in muscle mass and body composition often require consistent treatment over a longer period.
Which is better, testosterone C or E?
Testosterone cypionate and testosterone enanthate are clinically very similar. Both are long-acting testosterone esters that deliver the same active hormone once metabolized. Differences between them are minimal in practice, and the choice often depends on availability, physician preference, dosing schedule, and patient response rather than clear superiority of one over the other.
How often should I inject test E?
Testosterone enanthate is commonly administered every 1 to 2 weeks, depending on the treatment protocol. Many clinicians prefer weekly or split dosing to maintain more stable testosterone levels and reduce fluctuations. The optimal injection frequency should be determined based on individual response and laboratory monitoring.
Is 200mg of test E enough to build muscle?
A dose of 200 mg per week may increase testosterone levels and potentially support muscle development, particularly in individuals with low baseline testosterone. However, responses vary significantly between individuals, and muscle growth also depends on training, nutrition, and overall health. In clinical settings, dosing is aimed at restoring physiological levels rather than maximizing muscle gain, and treatment should always be guided by a healthcare professional.
Share this page:
- Share on X (Opens in new window) X
- Share on Facebook (Opens in new window) Facebook
- Email a link to a friend (Opens in new window) Email
- Share on LinkedIn (Opens in new window) LinkedIn
- Share on Reddit (Opens in new window) Reddit
- Share on Pinterest (Opens in new window) Pinterest
- Share on Telegram (Opens in new window) Telegram
- Share on WhatsApp (Opens in new window) WhatsApp
- Share on Tumblr (Opens in new window) Tumblr
Written by Steroids USA
Pay with WISE APP or Remitly
Pay with WISE App or Remitly
Fast money transfers from USA for fast delivery of steroids
Secure delivery in USA
100% reliable shipping in USA
24x7 Support
Online 24 hours
Low cost delivery
Great shipping prices in USA
BULK ORDER DISCOUNT
If you are a reseller in the USA you can get a special DISCOUNT, we can give you up to 50% or more on bulk orders. If you want to make a bulk order, we can negociate for orders of over USD$4,000, contact us by email.
Steroids info













Leave a Reply