Category
- Best Peptides for muscle growth
- Geno Pharma Domestic Warehouse 2 (Canada&USA)
- GPH(Domestic Shipping US) Warehouse 1
- Human Pharma Premium
- Phar Labs Premium-Select
- Steroids on Sale USA, Real Steroids Online
- New arrivals in USA
- Most popular steroids in USA
- Antiestrogens / Gonadotropins
- Bangkok Steroid USA
- Biopharma Steroid USA
- British Dragon
- Anabolic Steroids for Horses
- Fat-burners
- Gen Pharma USA
- Medical Pharma Steroid USA
- Medical Tech Steroid USA
- Novocrine Steroids
- HGH USA
- Omega Labs Steroid USA
- Rotterdam Steroids USA
- SARMs USA
- Sciroxx
- Sydgroup Steroid USA
- Big vetenary Steroid USA
- Watson Steroids
- XT Labs Steroids
Most Popular steroids USA
-
 Trenbolone 200 20ml - Sydgroup
$53.00
Trenbolone 200 20ml - Sydgroup
$53.00 -
 Primobolan for sale 100mg 10 ml Human Pharma in USA
$115.00
Primobolan for sale 100mg 10 ml Human Pharma in USA
$115.00 -
 NPP Steroid 100mg 10 ml Premium Domestic USA
NPP Steroid 100mg 10 ml Premium Domestic USA
$99.00Original price was: $99.00.$80.00Current price is: $80.00. -
 Anavar for Sale in USA – 10mg 80 Tabs GPH-Premium
Anavar for Sale in USA – 10mg 80 Tabs GPH-Premium
$110.00Original price was: $110.00.$80.00Current price is: $80.00. -
 Buy Anavar 10mg – Purchase Geno Pharma
$99.00
Buy Anavar 10mg – Purchase Geno Pharma
$99.00 -
 Testosterone Cypionate Buy 300mg 10ml Geno Pharma
$99.00
Testosterone Cypionate Buy 300mg 10ml Geno Pharma
$99.00 -
 Buy Testosterone E 300mg 10 ml Geno Pharma Domestic USA/CA
$99.00
Buy Testosterone E 300mg 10 ml Geno Pharma Domestic USA/CA
$99.00 -
 Testosterone Cypionate 200 Biopharma 10 amp
Testosterone Cypionate 200 Biopharma 10 amp
$99.00Original price was: $99.00.$72.00Current price is: $72.00. -
 Testosterone 400 Biopharma 10 Ampoules
Testosterone 400 Biopharma 10 Ampoules
$99.00Original price was: $99.00.$75.00Current price is: $75.00. -
 Eq 300 steroid Rotterdam 10ml
Eq 300 steroid Rotterdam 10ml
$79.00Original price was: $79.00.$69.00Current price is: $69.00. -
 Steroid Deca Geno Pharma 300mg 10ml
Steroid Deca Geno Pharma 300mg 10ml
$110.00Original price was: $110.00.$99.00Current price is: $99.00. -
 Boldenone Cypionate 200 mg / 10 mL Geno Pharma
Boldenone Cypionate 200 mg / 10 mL Geno Pharma
$90.00Original price was: $90.00.$85.00Current price is: $85.00.

Steroid Deca Geno Pharma 300mg 10ml
$110.00 Original price was: $110.00.$99.00Current price is: $99.00.
Steroid Deca Geno Pharma 300mg 10ml refers to a nandrolone decanoate formulation commonly encountered in clinical, regulatory, and non-medical contexts. Nandrolone decanoate is a long-acting anabolic–androgenic steroid with documented medical applications in selected conditions such as anemia, osteoporosis, and severe catabolic states, as well as a well-established risk profile. Its pharmacology is characterized by depot-based intramuscular release, prolonged systemic exposure, and significant endocrine effects, requiring careful patient selection, monitoring, and risk mitigation. Outside supervised medical use, exposure is associated with cardiovascular, endocrine, and metabolic complications, underscoring the importance of clinical oversight and evidence-based evaluation.
Share this page:
- Share on X (Opens in new window) X
- Share on Facebook (Opens in new window) Facebook
- Email a link to a friend (Opens in new window) Email
- Share on LinkedIn (Opens in new window) LinkedIn
- Share on Reddit (Opens in new window) Reddit
- Share on Pinterest (Opens in new window) Pinterest
- Share on Telegram (Opens in new window) Telegram
- Share on WhatsApp (Opens in new window) WhatsApp
- Share on Tumblr (Opens in new window) Tumblr
Steroid Deca (Nandrolone Decanoate): Medical Uses, Benefits, and Side Effects
Deca Durabolin is the popular trade name for nandrolone decanoate, an anabolic steroid originally developed for medical use. Medically, nandrolone has been used to treat conditions like anemia, osteoporosis, and muscle wasting. However, it is also infamously misused by athletes and bodybuilders to accelerate muscle growth. Below, we provide a science-based overview of Deca Durabolin – what it is, how it’s used (injection form), the benefits or results one might expect, the side effects and health risks, as well as its legal status and cost. All information is drawn from recent medical and scientific sources.
What Is Steroid Deca (Nandrolone Decanoate)?
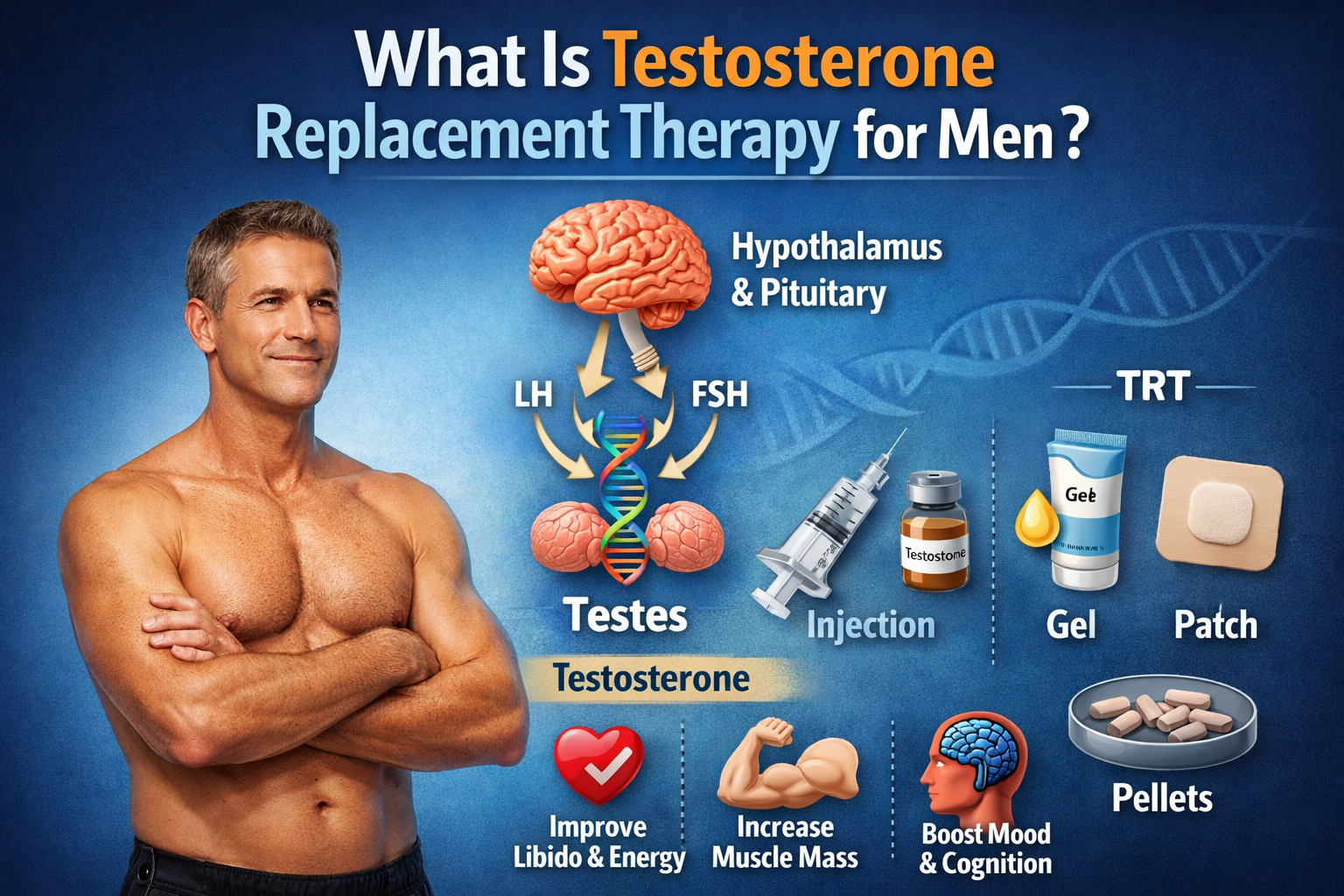
Deca Durabolin is the brand name for nandrolone decanoate, a synthetic anabolic-androgenic steroid (AAS) derived from testosterone. In essence, nandrolone is a laboratory-made version of the male hormone testosterone, modified to enhance muscle-building (anabolic) effects while reducing male-sex-hormone (androgenic) effects (cleveland) (pmc.ncbi.nlm). It was initially created for legitimate medical therapies:
Medical uses: Doctors have prescribed nandrolone for certain types of anemia (to stimulate red blood cell production) and to treat osteoporosis in postmenopausal women. It has also been used for muscle-wasting conditions (such as HIV/AIDS-related wasting) and even breast cancer palliation. These uses leverage nandrolone’s ability to promote tissue growth and increase bone density.
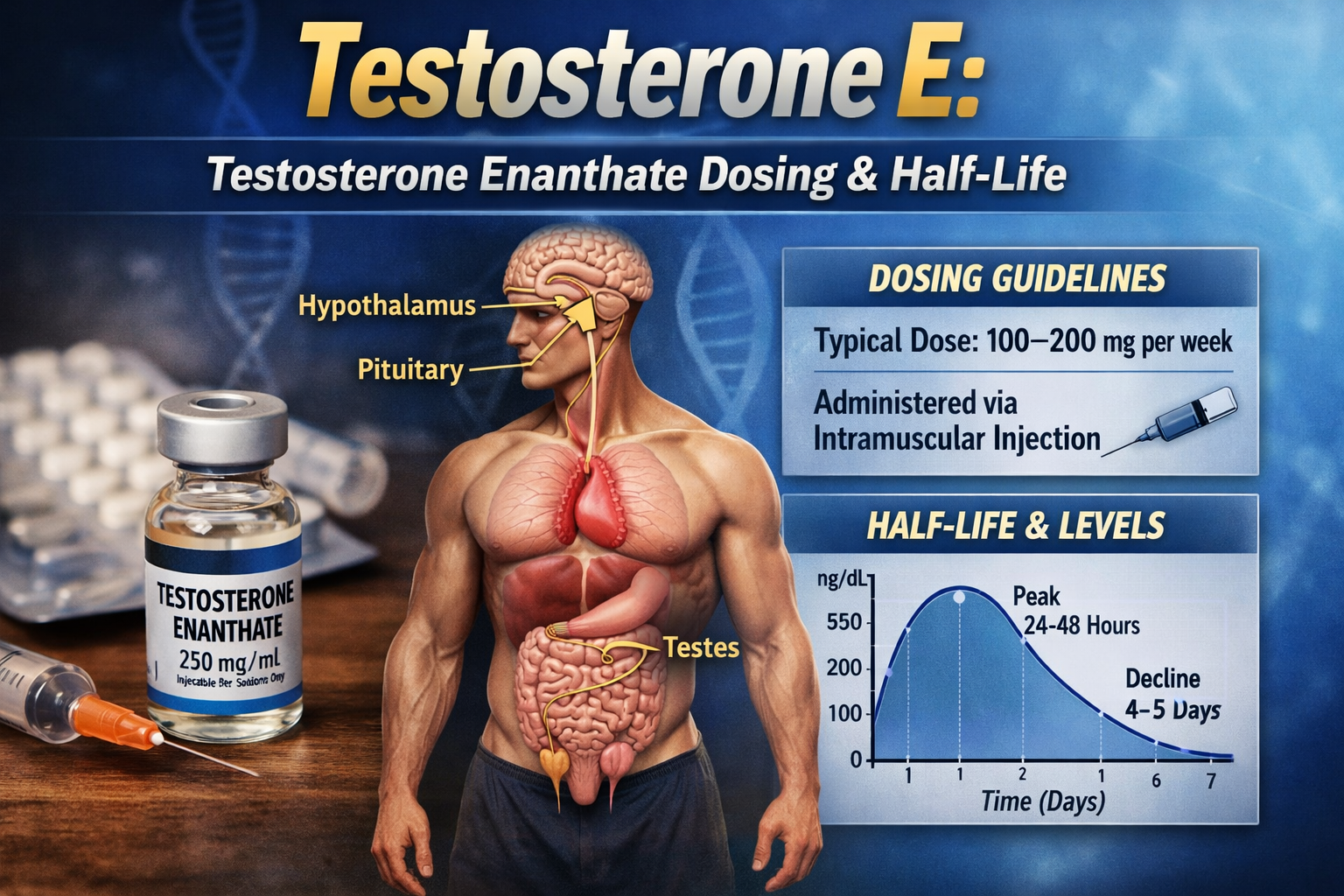
How it works: Nandrolone decanoate is typically administered as an intramuscular injection. The steroid ester “decanoate” slows its release, giving it a long half-life of ~6–12 days and a duration of action around 2–3 weeks per injectionen.wikipedia.org. Once in the body, nandrolone acts like a weaker form of testosterone, binding to androgen receptors in muscle and bone. It strongly stimulates anabolic processes (such as protein synthesis and red blood cell formation) with relatively less androgenic (masculinizing) activity. This high anabolic-to-androgenic ratio is one reason for its medical appeal and its popularity among bodybuilders.
Despite legitimate uses, Deca Durabolin gained notoriety because of off-label misuse in sports. Some athletes and bodybuilders began taking it in high doses to “bulk up” and improve performance – practices that are illegal and unsafe. In the United States and many countries, nandrolone is a controlled substance (Schedule III in the U.S.), meaning using or possessing it without a prescription is illegal. In fact, no FDA-approved pharmaceutical company currently distributes Deca Durabolin in the U.S., so any product on the market is likely imported or illicit. Both the NCAA and the World Anti-Doping Agency (WADA) explicitly ban nandrolone for athletes.
Pharmacology of Nandrolone Decanoate (Steroid Deca “Geno Pharma 300mg 10ml”)
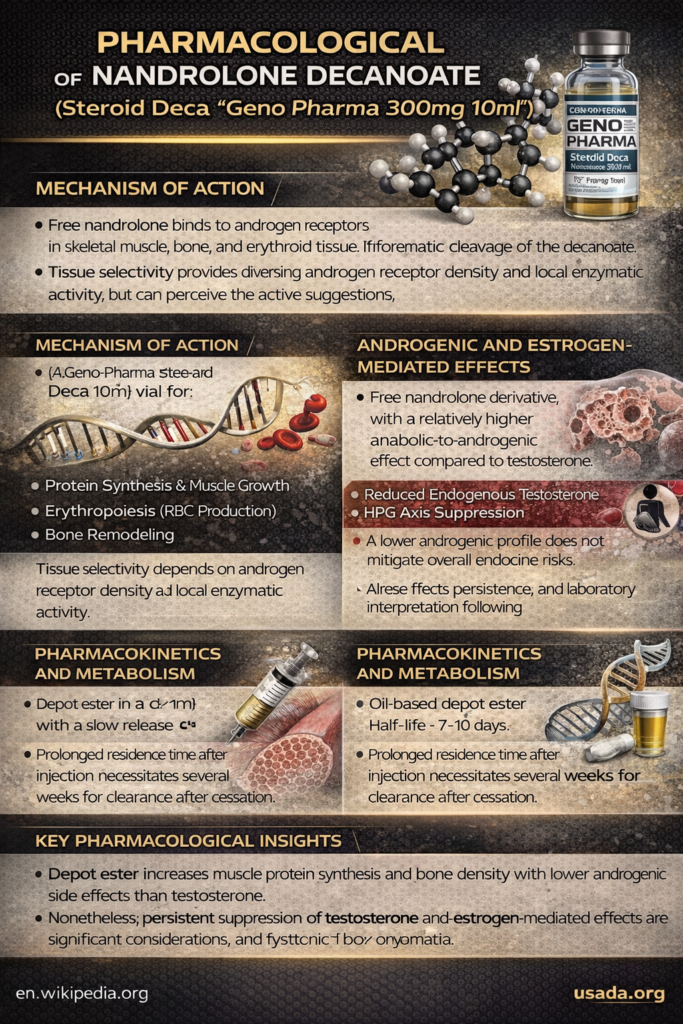
In clinical, academic, and forensic settings, products marketed as “Steroid Deca” Geno Pharma 300mg 10ml are intended to contain nandrolone decanoate as the active compound. From a medical perspective, it is essential to distinguish between commercial labeling or concentration claims (such as 300 mg per 10 mL) and the true pharmacological behavior, which is determined exclusively by the active molecule and its ester-mediated release characteristics.
The pharmacological principles described below apply to nandrolone decanoate regardless of manufacturer, brand name, or vial presentation.
Mechanism of Action of Nandrolone Decanoate
Androgen Receptor Signaling and Tissue Selectivity
Following intramuscular administration and enzymatic cleavage of the decanoate ester, free nandrolone binds to the androgen receptor (AR). This receptor–ligand interaction initiates transcriptional signaling pathways involved in protein synthesis, muscle fiber hypertrophy, erythropoiesis, and bone remodeling.
Tissue selectivity arises from differences in androgen receptor density, intracellular co-regulator proteins, and local enzymatic activity, contributing to interindividual variability in both therapeutic response and adverse effects.
Anabolic vs Androgenic Effects: Clinical Interpretation
Nandrolone is often described as having a relatively higher anabolic-to-androgenic activity compared with testosterone. Clinically, this distinction should be interpreted with caution. A comparatively lower androgenic profile does not imply a reduced overall risk.
The clinical expression of anabolic and androgenic effects is influenced by dose, exposure duration, baseline endocrine status, and tissue-specific metabolism, all of which determine both desired outcomes and toxicity.
Progestogenic Activity and Its Clinical Relevance
As a 19-nortestosterone derivative, nandrolone exerts endocrine effects that extend beyond classical androgen receptor activation. Compounds in this structural class are associated with suppression of the hypothalamic–pituitary–gonadal (HPG) axis, resulting in decreased gonadotropin release and reduced endogenous testosterone production.
Clinically, this mechanism contributes to sexual dysfunction, infertility, and prolonged hypogonadism observed after exposure to nandrolone-containing preparations.
Aromatization and Estrogen-Mediated Effects
Although nandrolone undergoes aromatization at a lower rate than testosterone, estrogen-mediated effects remain clinically relevant, particularly with sustained systemic exposure. These effects include fluid retention and gynecomastia and reflect downstream interactions within the broader endocrine network.
Pharmacokinetics and Metabolism
Depot Release After Intramuscular Injection
Nandrolone decanoate is formulated as an oil-based intramuscular depot. The long decanoate ester delays systemic release, making depot diffusion the rate-limiting step in drug availability. After gradual release from muscle tissue, ester hydrolysis liberates active nandrolone into circulation.
Half-Life, Time-to-Peak, and Duration of Action
The pharmacokinetic profile of nandrolone decanoate is characterized by prolonged exposure due to slow depot release, followed by faster systemic clearance of free nandrolone. Clinically, this results in biological activity that may persist for several weeks after a single injection.
This prolonged duration has important implications for therapeutic monitoring, adverse-effect persistence, and laboratory interpretation following discontinuation.
Metabolites and Laboratory Detection Implications
Nandrolone metabolism produces characteristic urinary metabolites, most notably 19-norandrosterone (19-NA) and 19-noretiocholanolone (19-NE). These metabolites are central to clinical toxicology and anti-doping analyses.
Modern laboratory methodologies emphasize differentiation between endogenous trace production and exogenous administration, particularly in cases involving long-acting ester formulations such as nandrolone decanoate.
Formulations and Concentrations of Deca Durabolin (Steroid Deca)
Preparations marketed as Steroid Deca, including presentations such as Geno Pharma 300mg 10ml, are intended to deliver nandrolone decanoate as the active anabolic agent. From a clinical and regulatory standpoint, it is critical to distinguish between formulation type, concentration labeling, and manufacturing quality, as these factors influence pharmacological reliability, safety, and clinical interpretation.
Importantly, the pharmacodynamic properties of nandrolone decanoate remain constant across formulations; however, formulation quality and concentration accuracy can vary substantially depending on the source.
Pharmaceutical vs Non-Pharmaceutical Preparations
Pharmaceutical-grade nandrolone decanoate preparations are produced under regulated manufacturing standards, ensuring validated concentration, sterility, and stability. These products are typically supplied in standardized ampoule strengths (e.g., 25 mg, 50 mg, or 100 mg per mL) and are accompanied by regulatory oversight, batch testing, and pharmacovigilance reporting.
In contrast, non-pharmaceutical or non-regulated preparations—often encountered in products labeled as Steroid Deca—are not subject to the same quality controls. Clinical literature and forensic analyses consistently report variability in actual nandrolone content, sterility assurance, and excipient composition in such products. This variability introduces uncertainty in both exposure estimation and adverse-effect risk.
From a medical perspective, the absence of regulatory oversight complicates dose interpretation, increases the likelihood of contamination or mislabeling, and may exacerbate endocrine and cardiovascular risks associated with androgen exposure.
Concentration Variability (e.g., 300 mg / 10 mL)
Concentration descriptors such as “300 mg / 10 mL”, commonly associated with labels like Geno Pharma 300mg 10ml, represent the claimed total amount of nandrolone decanoate per vial, rather than a standardized pharmaceutical dosing unit.
In clinical terms, higher-concentration multi-dose vials raise several considerations:
Accuracy of concentration may vary across batches in non-regulated products.
Higher total drug content per container increases the risk of unintentional supraphysiologic exposure.
Interpretation of patient-reported use becomes more complex when concentration labeling does not align with approved pharmaceutical standards.
For clinicians and researchers, concentration variability underscores the importance of focusing on biological exposure and observed clinical effects, rather than relying solely on labeled milligram claims when evaluating androgen-related outcomes.
Stability, Storage, and Quality Considerations
Nandrolone decanoate is formulated as an oil-based injectable solution, which confers chemical stability when manufactured and stored appropriately. Pharmaceutical preparations are designed to maintain stability under defined storage conditions, typically protected from excessive heat, light, and microbial contamination.
In non-pharmaceutical contexts, however, stability and quality may be compromised by:
Inconsistent excipient composition
Inadequate sterilization processes
Improper storage conditions prior to distribution
Degradation of the active compound or contamination of the oil vehicle can increase the risk of injection-site reactions, systemic inflammatory responses, and unpredictable pharmacokinetics. From a safety standpoint, these factors further differentiate regulated medical use from unregulated exposure scenarios.
Clinical Relevance of Formulation Differences
For healthcare professionals, understanding formulation and concentration differences is essential when interpreting laboratory findings, assessing adverse effects, or evaluating patient histories involving Steroid Deca products.
Formulation-related uncertainty should always prompt a risk-based clinical approach, emphasizing patient safety, monitoring, and evidence-based counseling rather than assumptions of product equivalence.
Clinical and Therapeutic Uses of Deca Durabolin
From a medical standpoint, preparations marketed as Steroid Deca, including presentations such as Geno Pharma 300mg 10ml, are intended to deliver nandrolone decanoate, a synthetic anabolic–androgenic steroid with a long history of therapeutic investigation.
While non-medical use has shaped public perception, nandrolone decanoate was originally developed and studied for specific clinical indications, particularly in conditions characterized by catabolism, bone loss, or impaired erythropoiesis.
The sections below summarize established, historical, and emerging therapeutic contexts based on clinical evidence rather than anecdotal use.
Historical Indications and Current Clinical Evidence
Nandrolone decanoate was introduced into clinical practice in the mid-20th century, at a time when anabolic agents were actively explored for supportive treatment of chronic disease. Early indications focused on conditions involving negative nitrogen balance, reduced bone density, and impaired red blood cell production.
Over time, the clinical use of nandrolone declined in some regions due to regulatory changes, availability of alternative therapies, and concerns related to misuse. However, contemporary literature continues to reassess nandrolone’s risk–benefit profile in carefully selected patient populations, particularly in controlled, low-dose settings.
Current evidence emphasizes that therapeutic effects are context-dependent and require strict medical supervision, appropriate patient selection, and ongoing monitoring.
Anemia and Bone Marrow Failure Syndromes
One of the earliest and most studied clinical applications of nandrolone decanoate involves its role in stimulating erythropoiesis. Nandrolone enhances red blood cell production through androgen receptor–mediated effects on the bone marrow, leading to increases in hemoglobin and hematocrit.
Clinically, nandrolone has been investigated in:
Certain forms of anemia associated with chronic kidney disease
Bone marrow failure syndromes, including aplastic anemia
Situations where standard therapies are ineffective or contraindicated
Recent clinical discussions frame nandrolone as an adjunctive therapy, rather than a first-line agent, with careful attention to cardiovascular risk, hematologic response, and endocrine suppression.
Osteoporosis and Osteosarcopenia
Nandrolone decanoate has demonstrated clinically relevant effects on bone metabolism, including stimulation of osteoblastic activity and improvements in bone mineral density. Historically, this led to its investigation in postmenopausal osteoporosis and age-related bone loss.
More recently, interest has expanded toward osteosarcopenia, a condition characterized by the coexistence of low bone density and reduced muscle mass. In this context, nandrolone’s combined effects on bone strength and lean body mass are of particular interest.
Clinical research highlights that:
Improvements in bone density may occur alongside gains in muscle mass
Functional outcomes (strength, mobility) are as important as imaging-based endpoints
Long-term safety remains a critical limiting factor
Cachexia and Severe Catabolic States
Another important therapeutic context for nandrolone decanoate is the management of severe catabolic conditions, where involuntary weight loss and muscle wasting negatively affect prognosis and quality of life.
These conditions include:
Cancer-associated cachexia
Chronic inflammatory diseases
Advanced systemic illness with persistent negative nitrogen balance
In such settings, nandrolone has been explored for its ability to preserve or restore lean body mass, improve physical function, and support recovery when nutritional interventions alone are insufficient. Clinical interpretation increasingly emphasizes functional outcomes and patient-centered measures, rather than isolated changes in body weight.
Emerging Clinical Research (2023–2025)
Between 2023 and 2025, renewed scientific interest has focused on reevaluating nandrolone decanoate within modern clinical frameworks. Recent studies and ongoing trials have explored its potential role in:
Recovery from critical illness and prolonged immobilization
Treatment of osteosarcopenia in aging populations
Supportive therapy in selected endocrine or hematologic disorders
These investigations apply contemporary standards of clinical trial design, including defined endpoints, safety monitoring, and ethical oversight. Importantly, current research does not support broad or unsupervised use but instead seeks to clarify narrow, evidence-based indications where benefits may outweigh risks.
Clinical Perspective
For healthcare professionals, understanding the legitimate therapeutic contexts of nandrolone decanoate is essential when evaluating patient histories, interpreting laboratory findings, or managing complications associated with androgen exposure.
While products labeled as Steroid Deca are widely encountered outside regulated settings, the clinical value of nandrolone decanoate lies exclusively in controlled, medically supervised use, supported by evolving evidence rather than anecdotal outcomes.
Clinical Administration of Deca Steroid Injections
In medical and regulatory contexts, preparations marketed as Steroid Deca, including presentations such as Geno Pharma 300mg 10ml, are intended to deliver nandrolone decanoate via parenteral administration. Clinical administration principles focus on pharmacological rationale, patient safety, and regulatory compliance, rather than on non-medical dosing practices.
The following subsections outline how nandrolone decanoate has been approached in supervised clinical settings and how clinicians interpret administration-related risks.
Intramuscular Injection and Depot Pharmacology
Nandrolone decanoate is formulated as an oil-based intramuscular injection, designed to create a depot within muscle tissue. The long-chain decanoate ester slows systemic availability, resulting in prolonged and sustained androgen exposure following a single administration.
From a pharmacological perspective, depot formulations:
Reduce peak–trough fluctuations compared with short-acting esters
Prolong biological activity over days to weeks
Complicate rapid reversal of exposure if adverse effects occur
These properties underscore why intramuscular depot formulations require careful clinical consideration and are generally unsuitable for unsupervised or experimental use.
Dosing Principles in Medical Practice
In legitimate medical practice, nandrolone decanoate has historically been administered using low, infrequent doses tailored to specific indications and patient characteristics. Dosing decisions are guided by:
The therapeutic goal (e.g., hematologic support vs musculoskeletal outcomes)
Patient-specific factors such as age, sex, comorbidities, and baseline endocrine status
Ongoing assessment of benefit–risk balance
Contemporary clinical literature emphasizes that nandrolone, when used therapeutically, should be viewed as an adjunctive intervention, not a routine or first-line therapy. Importantly, supraphysiologic exposure markedly increases the risk of endocrine suppression, cardiovascular complications, and prolonged adverse effects.
Injection-Related Risks and Complications
Intramuscular administration of oil-based androgen preparations carries inherent risks, regardless of formulation source. Reported injection-related complications include:
Local pain, inflammation, or induration at the injection site
Sterile or infectious abscess formation
Vascular injury or accidental intravascular injection
Delayed hypersensitivity or inflammatory reactions
Non-pharmaceutical preparations further increase these risks due to uncertain sterility, excipient composition, and storage conditions. Clinically, unexplained injection-site symptoms should prompt evaluation for infection, inflammatory response, or product-related contamination.
Controlled Substance Handling and Documentation
Nandrolone decanoate is classified as a controlled substance in many jurisdictions, reflecting its potential for misuse and adverse health outcomes. In clinical environments, appropriate handling includes:
Secure storage consistent with controlled-drug regulations
Clear documentation of indication, administration, and patient consent
Ongoing monitoring and pharmacovigilance reporting
From a medico-legal standpoint, accurate documentation is essential when evaluating adverse events, laboratory abnormalities, or patient disclosures involving Steroid Deca products. Lack of verifiable sourcing or documentation significantly complicates clinical interpretation and risk management.
Clinical Perspective
The clinical administration of nandrolone decanoate requires an understanding of depot pharmacology, systemic persistence, and regulatory responsibility. While products labeled as Steroid Deca are widely encountered outside medical settings, the principles outlined above reflect best practices in supervised clinical contexts, where patient safety and evidence-based decision-making remain central.
Benefits and Expected Results of Nandrolone Decanoate
When evaluating products marketed as Steroid Deca, including presentations such as Geno Pharma 300mg 10ml, it is essential to frame benefits strictly within clinically supported contexts. Nandrolone decanoate has demonstrated measurable biological effects in selected medical scenarios; however, these effects must be distinguished from exaggerated or non-evidence-based claims frequently associated with non-medical use.
Medically Indicated Benefits
In supervised clinical settings, nandrolone decanoate has been associated with several therapeutically relevant effects, depending on indication and patient selection:
Improvement in nitrogen balance and lean body mass, particularly in catabolic states where muscle wasting contributes to functional decline
Stimulation of erythropoiesis, leading to increases in hemoglobin and hematocrit in certain anemia-related conditions
Positive effects on bone metabolism, including increases in bone mineral density in populations at risk for osteoporosis or osteosarcopenia
These benefits are not universal and are typically observed at carefully controlled doses, often as adjunctive therapy rather than standalone treatment. Clinical benefit should always be weighed against the risk of endocrine suppression and systemic adverse effects.
Clinical Outcomes and Measured Endpoints
Modern clinical research emphasizes objective, measurable endpoints when assessing the effects of nandrolone decanoate. Reported outcomes in the literature include:
Changes in lean body mass measured by imaging or body composition analysis
Bone mineral density assessed by dual-energy X-ray absorptiometry (DEXA)
Hematologic parameters, such as hemoglobin concentration and red blood cell count
Functional outcomes, including strength, mobility, and recovery metrics
Importantly, improvements in laboratory or imaging markers do not always translate into meaningful clinical benefit, particularly if functional capacity or quality of life is unchanged.
Limitations of Outcome Interpretation
Interpreting outcomes associated with nandrolone decanoate requires caution. Several factors limit the generalizability of reported benefits:
Many studies involve small sample sizes or short follow-up periods
Outcomes may be influenced by concurrent interventions, such as nutrition or physical rehabilitation
Adverse effects may emerge after the apparent therapeutic window
For clinicians, this underscores the need to avoid extrapolating isolated positive outcomes into broad treatment recommendations. The presence of biological effect does not inherently justify long-term or repeated exposure.
Contraindications and Patient Selection
Appropriate patient selection is central to the safe clinical use of nandrolone decanoate. Due to its systemic endocrine effects, contraindications are significant and non-negotiable in many populations.
Cardiovascular and Metabolic Risk Factors
Nandrolone decanoate is contraindicated or requires extreme caution in individuals with:
Pre-existing cardiovascular disease, including ischemic heart disease or cardiomyopathy
Uncontrolled hypertension
Dyslipidemia or other metabolic risk factors associated with atherosclerosis
Anabolic–androgenic steroids have been associated with adverse cardiovascular remodeling and increased thrombotic risk. In patients with underlying cardiovascular vulnerability, exposure may substantially increase morbidity.
Endocrine and Hormone-Sensitive Conditions
Due to its androgenic and endocrine-disrupting properties, nandrolone decanoate should be avoided in patients with:
Hormone-sensitive malignancies, including prostate or breast cancer
Pre-existing hypogonadism where further suppression of the hypothalamic–pituitary–gonadal axis would be harmful
Significant endocrine disorders affecting sex hormone regulation
In these contexts, androgen exposure may accelerate disease progression or lead to prolonged endocrine dysfunction.
Use in Adolescents, Pregnancy, and Lactation
Nandrolone decanoate is contraindicated in adolescents due to the risk of premature epiphyseal closure and irreversible effects on growth and sexual development.
Use during pregnancy or lactation is also contraindicated. Androgen exposure in these periods poses significant risks of fetal virilization, developmental abnormalities, and long-term reproductive consequences.
Clinical Perspective
The therapeutic potential of nandrolone decanoate exists only within narrow, carefully controlled clinical boundaries. For healthcare professionals, distinguishing evidence-based benefits from non-medical expectations is essential to ethical decision-making and patient safety.
Side Effects and Health Risks of Deca Durabolin Steroid
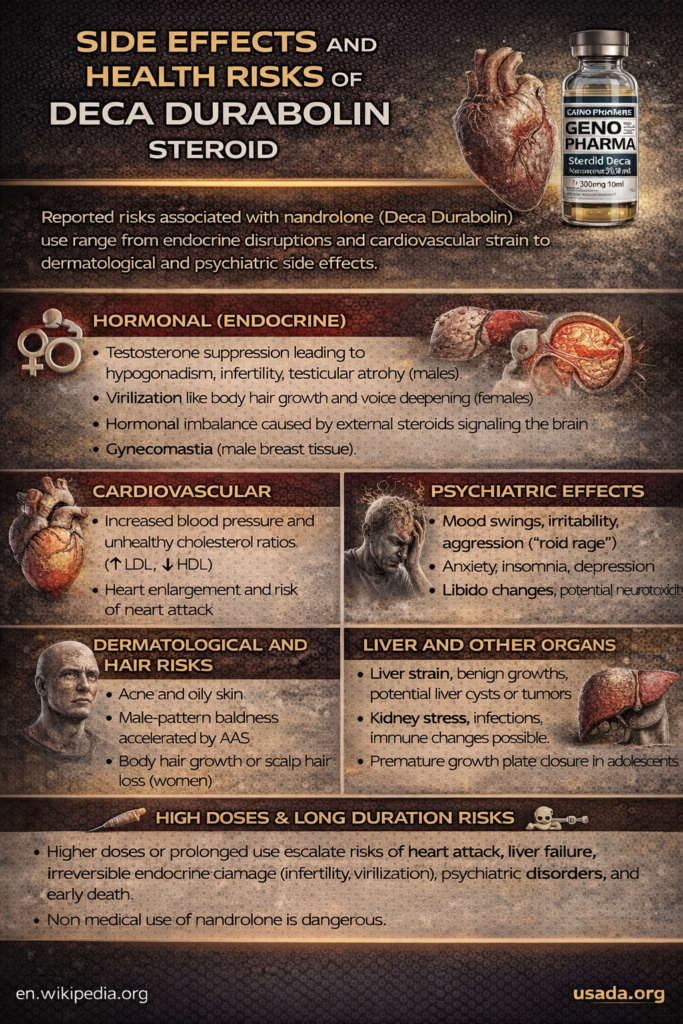
All anabolic steroids carry significant side effects, and nandrolone (Deca Durabolin) is no exception. The side effects can be broadly categorized into short-term effects that might be noticed while on the drug, and long-term or irreversible effects that develop with prolonged use or high doses. According to clinical research and reviews, the most frequently reported adverse effects of nandrolone involve the endocrine (hormonal), cardiovascular, dermatological, and psychiatric systems. Below is a breakdown of major side effects and risks:
Hormonal (Endocrine) disruptions: Nandrolone is a potent hormone, so it disrupts the body’s natural hormone balance. In males, Deca suppresses natural testosterone production, which can lead to testicular atrophy (shrinking testes), low sperm count (infertility), erectile dysfunction, and even breast development (gynecomastia) due to hormonal imbalances. These effects occur because external steroids send a signal to the brain to shut down gonadal testosterone production. The review of studies found virilization, gynecomastia, hormonal disorders, genital alterations, and infertility to be among the most common issues in steroid abuserspmc.ncbi.nlm.nih.gov. In females, even moderate nandrolone use can cause virilization – the development of male characteristics: for example, menstrual irregularities, enlargement of the clitoris, excessive body hair growth, male-pattern baldness, a deeper voice, and changes in libido (cleveland)(pmc.ncbi). Some of these changes in women (like voice deepening or clitoral enlargement) can be permanent. Both men and women can experience libido changes and mood disturbances as hormones swing abnormally.
Psychiatric effects: Anabolic steroids including Deca Durabolin can affect the mind. Users may experience mood swings, irritability, aggression (“roid rage”), or depression. Some develop anxiety or insomnia as well. These neuropsychiatric side effects are due to the steroid’s impact on neurotransmitters and hormone levels in the brain. Cases of AAS abuse have been linked to increased aggression and even violent behavior in certain individuals, as well as depressive episodes when coming off the drug.
Skin and hair: Nandrolone’s androgenic effects, though lower than testosterone’s, can still cause acne and oily skin, as well as accelerated baldness in those predisposed (male-pattern hair loss). Oily skin and severe acne (especially on the back or shoulders) are a common complaint during steroid cycles. On the flip side, women might notice male-pattern hair issues like facial hair growth or thinning scalp hair due to virilization.
Cardiovascular: Perhaps the most insidious risks are to the heart and blood vessels. Anabolic steroids can raise blood pressure and alter cholesterol levels (typically decreasing “good” HDL cholesterol and raising “bad” LDL). Nandrolone abuse has been associated with arterial damage, hypertension, and abnormal clotting. Over time, this contributes to atherosclerosis. The Cleveland Clinic notes nandrolone can lead to hardening of the arteries and plaque buildup, increasing risk of heart attack or stroke. Moreover, steroids may enlarge the heart muscle (left ventricular hypertrophy), reducing cardiac function and potentially leading to heart failure. Indeed, even at therapeutic doses, cases of cardiomyopathy (heart muscle damage) and heart failure have been reported with nandrolone.
Liver: Oral anabolic steroids are known for liver toxicity, but injectable nandrolone is not C-17 alpha alkylated, so it’s less harsh on the liver than, say, Dianabol or Winstrol. Nonetheless, high-dose or long-term nandrolone use can still damage the liver. It has been linked to peliosis hepatis (blood-filled cysts in the liver) and benign liver tumors, which sometimes can turn malignant. These liver lesions often have no symptoms until they are advanced, and they can be life-threatening if they cause liver failure or hemorrhage. Liver enzymes may become elevated as a sign of hepatic strain during steroid use.
Other organ systems: Kidney stress can occur (especially if blood pressure rises or if counterfeit products contain contaminants). Some reports have noted impaired kidney function or even specific nephrotoxic effects with long-term AAS use. Musculoskeletal problems can include an increased risk of tendon injuries – muscles get stronger quickly, but tendons may lag behind, leading to tears. Steroids can also prematurely stop bone growth in adolescents, stunting height permanentlymy.clevelandclinic.org (hence they should never be used by anyone who hasn’t finished growing). Immune system changes and even a predisposition to infections (due to hormonal stress) are also possible.
It’s important to realize that many of these side effects are dose-dependent. Using moderate medical doses for a short duration might cause mild, reversible effects like acne or transient mood changes. But high doses or prolonged use (the kind seen in steroid abuse) dramatically elevate the risks and can lead to permanent harm or even death. For example, chronic steroid users have suffered fatal heart attacks in their 30s-40s, and autopsies often reveal enlarged hearts and advanced arterial plaque. Psychiatric effects have led to suicides in some AAS abusers. Endocrine damage (like long-term hypogonadism in men or infertility) can require years of treatment to recover, if recovery happens at allpmc.ncbi.nlm.nih.gov. Because nandrolone is a “sex hormone” analog, it touches nearly every system in the body, meaning no organ is completely spared from potential adverse effectspmc.ncbi.nlm.nih.gov.
Bottom line: The side effect profile of Deca Durabolin is extensive, ranging from cosmetic annoyances (acne, hair loss) to life-threatening conditions (heart attack, liver failure). A systematic review concluded that the endocrine disturbances (like hormonal chaos, virilization, infertility) were the most frequently reported issues, followed by cardiovascular dysfunctions, skin disorders, and psychiatric disorders among nandrolone abusers. Everyone’s tolerance differs, but no one is immune to these effects when the dose is high enough for long enough. This underscores why medical supervision is critical for anyone receiving nandrolone and why non-medical use is so dangerous.
Safety Monitoring and Risk Mitigation
In any legitimate medical context involving nandrolone decanoate, including cases where patients report exposure to products marketed as Steroid Deca (e.g., Geno Pharma 300mg 10ml), structured safety monitoring is essential. Due to the drug’s systemic endocrine effects, long biological persistence, and dose-dependent toxicity, proactive risk mitigation is a cornerstone of responsible clinical management.
This section outlines the core principles used in supervised settings to minimize adverse outcomes and support evidence-based decision-making.
Baseline Evaluation Prior to Therapy
Before initiating nandrolone decanoate therapy, a comprehensive baseline evaluation is required to identify contraindications, establish reference values, and stratify patient risk. Core components typically include:
Detailed medical history, with emphasis on cardiovascular disease, thromboembolic events, endocrine disorders, and hormone-sensitive malignancies
Baseline cardiovascular assessment, including blood pressure and risk factor profiling
Endocrine evaluation, particularly gonadal axis function and prior androgen exposure
Baseline laboratory testing, to document pre-treatment hematologic, metabolic, and hepatic status
Establishing accurate baseline data is critical, as many adverse effects associated with nandrolone decanoate are relative changes from individual baseline values, rather than absolute laboratory thresholds.
Laboratory Monitoring During Treatment
Ongoing laboratory surveillance allows early detection of adverse physiological changes and informs timely intervention. In clinical practice, monitoring focuses on organ systems most affected by anabolic–androgenic steroids:
Hematologic parameters, including hemoglobin and hematocrit, to detect excessive erythrocytosis or hyperviscosity risk
Lipid profile, with particular attention to reductions in high-density lipoprotein (HDL) cholesterol and elevations in low-density lipoprotein (LDL) cholesterol
Liver function markers, even though nandrolone is not a C-17 alpha–alkylated steroid, as hepatic stress has been reported with prolonged exposure
Endocrine markers, to assess suppression of the hypothalamic–pituitary–gonadal axis and downstream hormonal effects
The long-acting depot nature of nandrolone decanoate necessitates extended monitoring, as laboratory abnormalities may persist or evolve weeks after administration.
Criteria for Dose Adjustment or Discontinuation
Clinical decision-making regarding continuation, modification, or cessation of nandrolone decanoate therapy is guided by risk–benefit reassessment, rather than isolated laboratory changes alone.
Common clinical triggers for dose adjustment or discontinuation include:
Progressive hematocrit elevation suggestive of increased thrombotic risk
Clinically significant deterioration of lipid profile
Evidence of cardiovascular strain, such as worsening hypertension or cardiac symptoms
Persistent or worsening endocrine suppression, particularly when recovery is unlikely with continued exposure
Emergence of neuropsychiatric or systemic adverse effects that outweigh therapeutic benefit
Given the prolonged pharmacokinetics of nandrolone decanoate, discontinuation does not result in immediate resolution of effects. Clinicians must anticipate delayed normalization and continue monitoring beyond the cessation of therapy.
Clinical Perspective
Effective safety monitoring transforms nandrolone decanoate from a high-risk hormone into a carefully controlled therapeutic agent within narrow clinical boundaries. For healthcare professionals, structured monitoring and early risk mitigation are essential not only for patient safety, but also for ethical and medico-legal accountability when managing exposure to Steroid Deca products in both therapeutic and non-therapeutic contexts.
Drug–Drug Interactions and Clinical Context
In clinical practice, nandrolone decanoate does not exist in isolation. Patients exposed to androgenic agents, including products marketed as Steroid Deca (e.g., Geno Pharma 300mg 10ml), often present with concomitant medications or underlying conditions that significantly influence risk, interpretation of laboratory findings, and clinical decision-making.
Understanding potential drug–drug interactions is essential for preventing avoidable adverse outcomes and misattribution of clinical findings.
Interactions With Anticoagulants
Nandrolone decanoate may alter hemostatic balance through its effects on erythropoiesis, hematocrit, and platelet activity. When combined with anticoagulant therapy, particularly vitamin K antagonists, androgen exposure may increase the risk of bleeding complications or unstable anticoagulation control.
Clinically, unexplained changes in coagulation parameters or thrombotic events should prompt evaluation for androgen exposure, including non-prescribed sources.
Concomitant Use With Corticosteroids
Concurrent exposure to nandrolone decanoate and systemic corticosteroids may amplify fluid retention, electrolyte imbalance, and cardiovascular strain. Both drug classes influence sodium and water homeostasis, potentially increasing the risk of edema, hypertension, and cardiac workload.
In patients receiving long-term corticosteroid therapy, additional androgen exposure complicates assessment of musculoskeletal outcomes, as observed changes in muscle mass or bone density may reflect overlapping pharmacologic effects rather than true disease modification.
Risks Associated With Other Androgens or Anabolic Agents
Combined exposure to multiple androgens—whether prescribed or illicit—substantially increases the risk of endocrine suppression, cardiovascular toxicity, and neuropsychiatric effects. Stacking androgenic agents obscures dose–response relationships and complicates attribution of adverse effects.
From a clinical standpoint, patients may underreport non-prescribed androgen use, leading to misinterpretation of symptoms, laboratory abnormalities, or treatment failure. A high index of suspicion is therefore warranted in cases of unexplained hormonal or cardiovascular disturbances.
Diagnostic and Laboratory Confounding
Nandrolone decanoate can significantly interfere with laboratory interpretation, particularly in endocrine and cardiovascular assessments. Suppression of endogenous testosterone production, altered lipid profiles, and changes in hematologic parameters may be misattributed to primary disease processes if androgen exposure is not disclosed.
Additionally, long-acting ester formulations may produce persistent laboratory abnormalities long after discontinuation, complicating longitudinal assessment and follow-up. In athletic or occupational health settings, nandrolone metabolites may also confound toxicology or doping analyses.
Clinical Perspective
Drug–drug interactions and diagnostic confounding represent a critical, yet often underappreciated, aspect of nandrolone decanoate exposure. For healthcare professionals, recognizing these interactions ensures more accurate diagnosis, safer pharmacologic management, and appropriate risk mitigation when encountering patients with known or suspected exposure to Steroid Deca products.
Legality and Price of Deca Durabolin
Legal status: Nandrolone decanoate (Deca Durabolin) is a controlled substance in most countries. In the United States, it is classified as a Schedule III controlled drug under the Anabolic Steroids Control Act. This means it’s illegal to buy, sell, or use Deca Durabolin without a valid prescription. Notably, although nandrolone remains FDA-approved for certain conditions, the original manufacturer (Organon) discontinued marketing Deca Durabolin in the U.S. around 2002, not due to safety issues but due to business reasons. As of 2023, there are no pharmaceutical companies distributing nandrolone in the U.S., so any product found domestically is either imported from abroad or produced in underground labs, both of which are illegal. Internationally, nandrolone is still available by prescription in countries such as Australia, the U.K., India, Belgium, and China. In those places it’s used medically in 25 mg, 50 mg or 100 mg ampoules for injection.
All major sporting bodies ban nandrolone. WADA (the World Anti-Doping Agency) and sports leagues impose strict penalties for athletes caught with nandrolone or its metabolites in their systemmy.clevelandclinic.org. Nandrolone can be detected in urine for a long time (months after use, in some cases) because its metabolites linger in the body fat and slow-release ester. Athletes have been stripped of titles for Deca Durabolin doping. So from both a legal and career standpoint, using this steroid can be ruinous.
Availability and price: The cost of Deca Durabolin can vary widely depending on the source:
In legitimate medical settings, where it’s available, Deca Durabolin is not a very expensive drug. For instance, in India (where Organon’s Deca-Durabolin is sold), a single 1 mL ampoule of 25 mg nandrolone decanoate costs around ₹279 (approximately $3-4 USD)healthpotli.com. In many developing countries, generic versions are inexpensive, reflecting the low manufacturing cost of this old drug.
On the black market or online steroid shops, Deca is usually sold in higher concentrations and larger vials. A common underground preparation is a 10 mL multi-dose vial containing 200 mg/mL (total 2000 mg of nandrolone). Such a vial might sell for on the order of $75 to $100 USD in the illicit market. For example, one online seller lists a 10 mL vial of 200 mg/mL nandrolone for $75, and higher concentration versions (e.g. 300 mg/mL) from certain brands can be around $100 or more. This puts the black-market cost roughly at $0.04–$0.05 per mg of steroid. Notably, these products are not regulated for quality, so purity and dosing may vary.
There is also a cost in terms of legality and health: Buying steroids illegally risks fraud (some “Deca” vials may not actually contain nandrolone, or could be contaminated). And if one is caught importing or distributing Deca Durabolin without a license, they could face legal penalties including fines or jail, which far outweigh the monetary price of the drug itself.
In summary, Deca Durabolin is relatively affordable as a substance – its notoriety is due to efficacy and risks, not high price. But obtaining it without a prescription is against the law in many places. If you see “Deca Durabolin for sale” online, remember that such sales are illicit. Always consider the hidden costs: potential legal troubles and the health “price” you might pay in side effects.
References: All information in this article is drawn from reputable medical and scientific sources, including the Cleveland Clinic Health Library (2023 review)my.clevelandclinic.org, peer-reviewed studies and reviews on nandrolone decanoatepmc.ncbi.nlm.nih.gov and official drug information. This ensures the content is up-to-date (2023–2025) and based on evidence. Always seek professional medical advice before considering any steroid use. The non-medical use of Deca Durabolin is dangerous and illegal in many regions – the short-term gains are not worth the long-term consequences to your health and legality. Stay informed and stay safe.
Author
Dr. Jonathan R. Keller, MD, PhD
Endocrinology & Clinical Pharmacology
Dr. Jonathan R. Keller is a physician–scientist specialized in endocrinology, anabolic–androgenic steroids, and clinical pharmacology, with over 15 years of experience in hormonal research and patient care. He holds a medical degree (MD) and a doctorate (PhD) in biomedical sciences, with academic training focused on androgen receptor signaling, steroid pharmacokinetics, and endocrine disruption.
Dr. Keller has contributed to peer-reviewed research on nandrolone decanoate, testosterone derivatives, and anabolic agents in clinical contexts including osteoporosis, sarcopenia, anemia, and hormone-related adverse effects. His work emphasizes risk stratification, laboratory monitoring, and evidence-based use of androgenic compounds in controlled medical settings.
He has served as a clinical consultant on drug safety, hormone misuse, and laboratory interpretation for healthcare institutions and has published educational content aimed at improving clinician awareness of anabolic steroid pharmacology and long-term health risks. His writing is grounded in current scientific literature (2023–2025) and adheres strictly to medical and ethical standards.
References:
Dave, B., Agarawal, S., Krishnan, A., Mayi, S., Rai, R. R., Dave, M. B., & Anil, A. (2025). Investigating the role of nandrolone decanoate in the management of osteosarcopenia in postmenopausal women: A prospective observational study. Cureus, 17(5), e84726. https://doi.org/10.7759/cureus.84726
URL:https://pmc.ncbi.nlm.nih.gov/articles/PMC12183668/Anstey, M., Litton, E., Habibi, M., Van der Lee, L., Palmer, R., Tran, N., Mammana, B., Scheepers, S., Palermo, A., Fiorilla, X., Mevavala, B., Regli, A., Jacques, A., & Wibrow, B. (2025). Muscle growth and anabolism in intensive care survivors (GAINS 2.0): Protocol for a multi-centre randomised; placebo controlled clinical trial of nandrolone in deconditioned adults recovering from critical illness. PLOS ONE, 20(2), e0315170. https://doi.org/10.1371/journal.pone.0315170
URL:https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0315170Fadah, K., Gopi, G., Lingireddy, A., Blumer, V., Dewald, T., & Mentz, R. J. (2023). Anabolic androgenic steroids and cardiomyopathy: An update. Frontiers in Cardiovascular Medicine, 10, 1214374. https://doi.org/10.3389/fcvm.2023.1214374
URL:https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2023.1214374/fullDi Fazio, N., Volonnino, G., Treglia, M., Delogu, G., Bubbico, T., Rinaldi, R., La Russa, R., & Maiese, A. (2025). Forensic approach in cases of anabolic-androgenic steroid abuse and cardiovascular mortality: Insights from autopsy, histopathology, immunohistochemistry and toxicology. Frontiers in Cardiovascular Medicine, 12, 1585205. https://doi.org/10.3389/fcvm.2025.1585205
URL:https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2025.1585205/fullvan Os, J., Smit, D. L., Bond, P., & de Ronde, W. (2025). Prolonged post-androgen abuse hypogonadism: Potential mechanisms and a proposed standardized diagnosis. Frontiers in Endocrinology, 16, 1621558. https://doi.org/10.3389/fendo.2025.1621558
URL:https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1621558/fullClé, D. V., Catto, L. F. B., Gutierrez-Rodrigues, F., Donaires, F. S., Pinto, A. L., Santana, B. A., Darrigo Jr., L. G., Valera, E. T., Koenigkam-Santos, M., Baddini-Martinez, J., Young, N. S., Martinez, E. Z., & Calado, R. T. (2023). Effects of nandrolone decanoate on telomere length and clinical outcome in patients with telomeropathies: A prospective trial. Haematologica, 108(5), 1300–1312. https://doi.org/10.3324/haematol.2022.281808
URL:https://haematologica.org/article/view/haematol.2022.281808/75197Nassani, M., El Fakih, R., Passweg, J., Cesaro, S., Alzahrani, H., Alahmari, A., Bonfim, C., Iftikhar, R., Albeihany, A., Halkes, C., Ahmed, S. O., Dufour, C., & Aljurf, M. (2023). The role of androgen therapy in acquired aplastic anemia and other bone marrow failure syndromes. Frontiers in Oncology, 13, 1135160. https://doi.org/10.3389/fonc.2023.1135160
URL:https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2023.1135160/fullChisari, M. G., Esposito, M., Alloca, S., Franco, S., Francaviglia, M., Volonnino, G., Rinaldi, R., Di Fazio, N., & Di Mauro, L. (2025). Anabolic–androgenic steroids and brain damage: A review of evidence and medico-legal implications. Forensic Sciences, 5(3), 31. https://doi.org/10.3390/forensicsci5030031
URL:https://www.mdpi.com/2673-6756/5/3/31Scendoni, R., Ricchezze, G., Mietti, G., Cerioni, A., Froldi, R., Cingolani, M., Buratti, E., & Cippitelli, M. (2024). Exogenous versus endogenous nandrolone in doping investigations: A systematic literature review. Applied Sciences, 14(22), 10641. https://doi.org/10.3390/app142210641
URL:https://www.mdpi.com/2076-3417/14/22/10641World Anti-Doping Agency. (2024). The World Anti-Doping Code: The 2024 Prohibited List (International Standard). (PDF).
URL:https://www.wada-ama.org/sites/default/files/2023-09/2024list_en_final_22_september_2023.pdfAmerican College of Cardiology. (2024, August 16). Expert’s approach to managing cardiovascular risk among athletes using anabolic androgenic steroids.
URL:https://www.acc.org/latest-in-cardiology/articles/2024/08/16/12/40/experts-approach-to-managing-cv-risk-among-athletes-using-aasMale sexual dysfunction and infertility associated with anabolic-androgenic steroid use: A systematic review and meta-analysis. (2024). Frontiers in Toxicology. https://doi.org/10.3389/ftox.2024.1422848
URL:https://www.frontiersin.org/journals/toxicology/articles/10.3389/ftox.2024.1422848/fullThe toxic effects of anabolic steroids “nandrolone decanoate” on cardiac and skeletal muscles with the potential ameliorative effects of silymarin and fenugreek seeds extract in adult male albino rats. (2023). BMC Pharmacology and Toxicology. https://doi.org/10.1186/s40360-023-00658-x
URL:https://link.springer.com/article/10.1186/s40360-023-00658-xFactors predicting normalization of reproductive hormones after cessation of anabolic-androgenic steroids in men: A single center retrospective study. (2023). European Journal of Endocrinology, 189(6).
URL:https://academic.oup.com/ejendo/article/189/6/601/7475314Anabolic-androgenic steroids are linked to depression and anxiety in weight-training men. (2024). Aging & Mental Health. https://doi.org/10.1080/13607863.2024.2300052
URL:https://www.tandfonline.com/doi/abs/10.1080/13607863.2024.2300052
Related products
Anastrozole 1mg 30 Pills Geno Pharma USA & CA- Domestic
$70.00Add to cartAnastrozole 1mg 30 Pills – Geno Pharma (USA & Canada Domestic Shipping)
Access pharmaceutical-grade Anastrozole 1mg tablets (30 count) from Geno Pharma with fast, reliable domestic shipping to both the United States and Canada. Orders containing only Geno Pharma products are dispatched directly from our North American distribution center, ensuring expedited delivery—typically within 1 to 3 business days in Canada and 3 to 5 business days across the U.S. via trusted local carriers.If your order includes products from other brands alongside Geno Pharma Anastrozole, it will be processed through our international fulfillment center, which may extend delivery times to 7–10 business days and involve additional customs clearance procedures.
With consistent stock availability, professional handling, and efficient logistics, Geno Pharma offers a dependable source for Anastrozole in North America—ideal for those seeking trusted hormone support with minimal wait time and no international shipping complications.
Clenbuterol tablet for Sale 10mcg 100 tabs Geno Pharma US/CA
$85.00Add to cartClenbuterol 10mcg Tablets (100 Count) – Available from Geno Pharma for US and Canadian Customers
Get fast and dependable access to Clenbuterol 10mcg tablets (100 units) through Geno Pharma, with domestic delivery options tailored for both the USA and Canada. When your order includes exclusively Geno Pharma Clenbuterol products, it will be shipped from our North American facility—ensuring quick arrival: typically 1 to 3 days within Canada and 3 to 5 days for U.S. destinations.Please note that combining Geno Pharma Clenbuterol with other brands in the same order will result in standard processing from a different fulfillment center, extending delivery times to 7–10 business days, with possible customs-related delays.
Our streamlined logistics ensure consistent stock availability and an efficient ordering process. For those prioritizing safety, speed, and dependable service, Geno Pharma remains a trusted source for Clenbuterol across North America.
Buy Anavar 10mg – Purchase Geno Pharma
$99.00Add to cartLooking to purchase Anavar online with fast, secure U.S. delivery? At SteroidsOnlineUSA, you can order authentic Anavar 10mg by Geno Pharma, one of the most trusted brands in the market. This high-quality Oxandrolone is available for sale in the USA with 3–5 day domestic shipping, no customs delays, and a 100% authenticity guarantee. To benefit from priority U.S. or Canada delivery, your order must include only Geno Pharma products. Mixed-brand orders follow standard shipping times (7–10 business days). For fastest results and a seamless purchase experience, choose Geno Pharma Anavar exclusively.
Tren Steroids – Tren Acetate 100mg 10ml Geno Pharma
$99.00Add to cartTrenbolone Acetate 100mg (10ml) by Geno Pharma is a powerful anabolic steroid widely used in the bodybuilding and athletic community for its fast-acting and highly potent effects on muscle growth, strength, and body composition. As a short-ester compound, Trenbolone Acetate is rapidly absorbed into the bloodstream, delivering noticeable performance benefits within just a few days of administration. It enhances nitrogen retention, increases red blood cell count, and boosts protein synthesis—key factors in promoting lean muscle mass and accelerated recovery. Often used during cutting or recomp cycles, Tren Acetate is favored for its ability to maintain muscle while reducing body fat. Due to its high androgenic potency, users should approach it with caution and be aware of potential side effects such as night sweats, increased aggression, or cardiovascular strain. Geno Pharma is known for producing high-purity injectable compounds, making this product a trusted option for athletes seeking reliable results.
Pay with WISE APP or Remitly
Pay with WISE App or Remitly
Fast money transfers from USA for fast delivery of steroids
Secure delivery in USA
100% reliable shipping in USA
24x7 Support
Online 24 hours
Low cost delivery
Great shipping prices in USA
BULK ORDER DISCOUNT
If you are a reseller in the USA you can get a special DISCOUNT, we can give you up to 50% or more on bulk orders. If you want to make a bulk order, we can negociate for orders of over USD$4,000, contact us by email.
Steroids info